Diagnosis of intracranial hemorrhages based on brain computed tomography with artificial intelligence
- Authors: Khoruzhaya A.N.1, Arzamasov K.M.1, Kodenko M.R.1, Kremneva E.I.1,2, Burenchev D.V.1
-
Affiliations:
- Research and Practical Clinical Center for Diagnostics and Telemedicine Technologies
- Russian Center of Neurology and Neurosciences
- Issue: Vol 6, No 2 (2025)
- Pages: 214-228
- Section: Original Study Articles
- Submitted: 13.01.2025
- Accepted: 05.02.2025
- Published: 08.07.2025
- URL: https://jdigitaldiagnostics.com/DD/article/view/645364
- DOI: https://doi.org/10.17816/DD645364
- EDN: https://elibrary.ru/RFYVMC
- ID: 645364
Cite item
Full Text
Abstract
BACKGROUND: Intracranial hemorrhages are associated with high mortality and risk of disability, requiring prompt and accurate diagnosis, particularly within the first 24 hours. The use of artificial intelligence technologies in analyzing brain computed tomography scans can shorten diagnostic time and improve diagnostic quality. The relevance of this study is emphasized by the limited number of certified artificial intelligence services for detecting intracranial hemorrhages in Russia and lacking data on their long-term effectiveness, highlighting the need for multicenter monitoring to assess the stability and accuracy of such systems in clinical practice.
AIM: The study aimed to assess the diagnostic accuracy and stability of an artificial intelligence service in detecting intracranial hemorrhages on non-contrast brain computed tomography scans in a multicenter clinical monitoring setting for 18 months.
METHODS: Anonymized brain computed tomography scans were used. The artificial intelligence service underwent a three-phase evaluation to evaluate its diagnostic accuracy and clinical performance using limited datasets. Two radiologists specializing in neuroimaging examined 80 brain computed tomography scans each month for 18 months, which had been preprocessed by the artificial intelligence service and randomly selected from the clinical workflow. The results were analyzed using ROC analysis with sensitivity, specificity, accuracy, and area under the curve.
RESULTS: During clinical monitoring, 1200 brain computed tomography scans were analyzed, with signs of intracranial hemorrhage detected in 48.3% of the scans. Based on the binary classification of intracranial hemorrhage presence or absence performed by the artificial intelligence service, the following diagnostic metrics were obtained: sensitivity, 97.4% (95.8–98.5); specificity, 75.4% (71.8–78.7); accuracy, 86.0% (83.9–87.9); and area under the curve, 94% (92.6–95.3). Eventually, a significant moderate positive correlation was observed in most diagnostic metrics and the time variable, except for sensitivity, which was affected by an update to the service version. However, full concordance between artificial intelligence-based markings and radiologist conclusions was noted in 28.5% of cases of identified intracranial hemorrhage, whereas discrepancies were found in 71.5%. The refined diagnostic metrics for cases with complete agreement with the radiologists’ report were as follows: sensitivity, 26.6%; specificity, 73.8%; accuracy, 50.1%; and area under the curve, 49.6%.
CONCLUSION: The current configuration of the artificial intelligence service allows ruling out intracranial hemorrhage with very high probability, which may be useful in the initial triaging of patients in emergency settings. However, low values of refined metrics indicate considerable discrepancies between radiologist reports and service-generated results regarding the interpretation of pathological findings.
Full Text
ОБОСНОВАНИЕ
Внутричерепное кровоизлияние (ВЧК) — потенциально опасное для жизни острое состояние, связанное с экстравазацией крови в ткани головного мозга, которое может возникнуть либо спонтанно, либо в случае травмы головы или хирургического вмешательства. Частота случаев ВЧК составляет более 25 на 100 тыс. человеко-лет по данным мировой статистики [1]. В Российской Федерации (РФ) ежегодно диагностируют геморрагический инсульт у 43 тыс. пациентов, он составляет от 10 до 15% общего числа случаев острых нарушений мозгового кровообращения [2]. Этиология нетравматического ВЧК разнообразна и включает артериальную гипертензию, инсульт, разрыв аневризмы, васкулопатию, тромбоз венозного синуса, артериовенозную фистулу, злокачественные новообразования, применение антикоагулянтов и реже — воспалительные заболевания [1]. ВЧК связано с высоким риском смерти (до 40–50% случаев) в первые сутки и инвалидизацией выживших пациентов [1, 2]. Обычно среди ВЧК различают эпидуральную, субдуральную, субарахноидальную и паренхиматозную (внутримозговую) локализации. Их дифференцируют с учётом клинической картины, особенностей визуализации и прогноза [2]. Компьютерная томография головного мозга (КТ ГМ) — основной диагностический инструмент, который применяют для обследования пациентов, поступающих в отделения неотложной помощи с головной болью или очаговыми неврологическими нарушениями [2]. Это относительно доступный и быстрый метод, результаты которого позволяют оценить тяжесть патологического процесса и определить тактику лечения [3]. Важно отметить, что диагностика ВЧК в первые 24 часа имеет решающее клиническое значение для снижения ранней смертности. Это достигают за счёт оценки вовлечённых областей мозга, объёма кровоизлияния, наличия высокорисковых признаков визуализации (симптомы «вихря», «пятна»), а также учёта клинического контекста, что позволяет своевременно определить тактику ведения пациента и организовать последующее наблюдение [4, 5].
Технологии искусственного интеллекта (ИИ) активно и успешно применяют в неотложной нейровизуализации для диагностики острого ишемического инсульта, нейроинфекций или компрессии спинного мозга [6]. ИИ востребован и для задач первичной диагностики ВЧК, поскольку данная патология имеет хорошо дифференцируемые диагностические признаки [7]. Целесообразность внедрения технологий ИИ для диагностики ВЧК обусловлена необходимостью ускорения (с 512 до 19 мин) [7] процесса первичной диагностики и повышения его точности [8], в частности за счёт сортировки пациентов и ранжирования рабочих списков рентгенологов [9, 10]. Однако применение ИИ в клинической практике возможно только при обеспечении высокой диагностической точности, которая определяется качеством обучающих и валидационных выборок [11], а также подтверждением полученных результатов на независимых внешних данных, что остаётся одной из ключевых проблем [12]. Одно из возможных её решений — предварительные и промежуточные проверки диагностических метрик на данных той медицинской информационной системы, где предполагают использовать сервис на основе технологий ИИ. Такие проверки позволяют проводить контроль качества для подтверждения диагностической эффективности сервисов ИИ с течением времени и дорабатывать их на основании регулярного анализа ошибок [13, 14]. Однако в зарубежной литературе представлено лишь ограниченное число исследований, посвящённых длительному наблюдению за жизненным циклом сервисов на основе ИИ для диагностики ВЧК с регулярной оценкой их эффективности, тогда как в отечественных источниках такие данные отсутствуют.
В Москве с 2020 года проводят эксперимент по использованию технологий компьютерного зрения для анализа медицинских изображений в интересах городской системы здравоохранения (Эксперимент), в котором задействованы более 50 сервисов на основе ИИ по 41 диагностическому направлению1. В РФ статусом «медицинское изделие» обладает 4 программных продукта на основе технологий ИИ, предназначенных для автоматического анализа цифровых медицинских изображений КТ ГМ на предмет наличия ВЧК. В апреле 2022 г. в Эксперименте начал работать первый такой диагностический сервис, который долгое время был единственным.
ЦЕЛЬ
Оценить диагностическую точность и устойчивость сервиса ИИ для диагностики ВЧК по данным нативной КТ ГМ в условиях многоцентрового клинического мониторинга на протяжении 18 мес.
МЕТОДЫ
Дизайн исследования
Проведён ретроспективный многоцентровой клинический мониторинг продолжительностью 18 мес.
Искусственный интеллект
Объект исследования — программное обеспечение ЦЕЛЬС® (ПО ЦЕЛЬС®, Россия) ООО «Медицинские скрининг системы» (ИИ-сервис). На этапе подачи заявки на участие в Эксперименте в апреле 2022 г. данный ИИ-сервис обучили на массиве из более 15 тыс. анонимизированных диагностических результатов исследования, полученных из двух медицинских учреждений. Их ранжировали по признаку «норма» и «патология» (ВЧК). В обучающую выборку включали все результаты КТ со следующими типами кровоизлияний:
- субдуральное;
- внутримозговое;
- эпидуральное;
- субарахноидальное.
Количество изображений «с патологией» для обучения составило 60% общего объёма выборки.
С целью определения исходных метрик точности работы ИИ-сервис прошёл предварительные клинико-технические испытания, что было необходимо для участия в Эксперименте. Для их проведения сформирован независимый набор данных, не использовавшийся в обучающей выборке, включающий 260 исследований КТ (КТ-исследование): 130 с выявленной патологией и 130 — с «нормой». На основе полученных данных сформировали таблицу с распределением исследований по типам диагностических результатов (ложноположительные, ложноотрицательные, истинно положительные и истинно отрицательные), после чего рассчитали метрики аналитической валидации. Получены следующие средние показатели диагностической точности: площадь под кривой (Area Under the Curve, AUROC) — 0,89; чувствительность — 0,84; специфичность — 0,74; точность — 0,79.
Входные данные (КТ-исследования ГМ) для обработки представлены в формате DICOM (Digital Imaging and Communications in Medicine). Результаты обработки включали:
- текстовое описание (DICOM SR);
- изображения с разметкой патологических областей (DICOM SC);
- числовую оценку вероятности наличия патологии в исследовании.
Результаты анализа, выполненного ИИ-сервисом, автоматически интегрированы и доступны вместе с исходными изображениями в Единой радиологической информационной системе (ЕРИС), которая входит в состав государственной информационной системы в сфере здравоохранения субъекта Российской Федерации — «Единая медицинская информационно-аналитическая система города Москвы» (ЕМИАС).
Для исследования использовали бинарную классификацию: определяли вероятность наличия патологии (ВЧК). Оценку её корректности в отношении отдельных типов кровоизлияний, а также точности сегментации и подсчёта объёмов в настоящем исследовании не проводили.
Тестирование сервиса на основе искусственного интеллекта
Согласно разработанной и апробированной в рамках Эксперимента методологии тестирования и мониторинга программного обеспечения на основе технологий ИИ [15], ИИ-сервис прошёл трёхэтапную проверку перед подключением к основному контуру ЕРИС ЕМИАС для обработки КТ-исследований.
- На этапе самотестирования успешно проведено тестирование технической совместимости алгоритма с обрабатываемыми данными.
- При функциональном тестировании оценивали полноту и достаточность инструментария ИИ-сервиса, а также возможность выполнения диагностической задачи.
- Калибровочное тестирование проводили для оценки клинической производительности и диагностических метрик сервиса.
На этапе функционального тестирования проверку ИИ-сервиса осуществляли с технической и клинической точки зрения, оценивали наличие и работоспособность его функционала в соответствии с базовыми диагностическими2 и функциональными требованиями3, которые разработали в рамках Эксперимента. Базовые диагностические требования включают обязательное и опциональное содержание ответа ИИ-сервиса, а также форму его представления. При решении клинической задачи диагностики ВЧК по данным КТ ГМ ИИ-сервис должен обязательно указывать вероятность наличия кровоизлияний в виде числового значения, тип/типы кровоизлияния (эпидуральное, субдуральное, субарахноидальное, внутримозговое), их объёмы (кроме субарахноидального), сегментировать зоны патологической плотности на самих исследованиях. Базовые функциональные требования определяют, какие изображения должен обрабатывать ИИ-сервис, а также содержательные и формальные характеристики предоставляемых результатов.
Этап калибровочного тестирования, следовавший за функциональным, направлен на подтверждение или опровержение заявленных разработчиком метрик эффективности работы ИИ-сервиса. Подсчитывали количество истинно положительных и отрицательных, ложноположительных и -отрицательных ответов в виде четырёхпольной таблицы, из чего рассчитывали основные метрики для оценки — AUROC, чувствительность, специфичность, точность, удельный вес ложноотрицательных и -положительных результатов. Кроме того, фиксировали минимальное, среднее и максимальное время анализа одного КТ-исследования. В качестве эталона в данной клинической задаче принято:
- значение AUROC — не менее 0,81;
- время, затрачиваемое на принятие, обработку изображения и передачу результатов анализа — не более 6,5 мин;
- удельный вес успешно обработанных исследований — не менее 90% [16, 17].
В течение 18 мес. провели суммарно три калибровочных тестирования с использованием набора данных, содержащего КТ-исследования с балансом классов 1:1. После каждого калибровочного тестирования ИИ-сервиса формировали протокол, в который входили информация о его наименовании, типе, компании-поставщике, а также данные об обработанных изображениях, полученные метрики и решение о том, соответствует ли данный ИИ-сервис принятым эталонным значениям для допуска к дальнейшей работе в ЕРИС ЕМИАС.
Самотестирование проводили на предоставленных в свободном доступе обезличенных диагностических изображениях в формате DICOM с приложением файла в формате Exсel, где дополнительно были указаны модальность, тип диагностической процедуры, производитель и модель диагностического устройства. Функциональное и калибровочное тестирования проводили с использованием эталонного набора данных MosMedData4. Для функционального тестирования использовали 5 КТ-исследований (с патологией, «нормой» и артефактом — 2, 2, 1 соответственно), для калибровочного — 100 КТ-исследований (по 50 с патологией и без).
Клинический мониторинг
ИИ-сервис подключили на потоковую обработку КТ-исследований ГМ 28.04.2022 из 56 медицинских организаций стационарной медицинской помощи. Данные о результатах их обработки собирали с апреля 2022 по сентябрь 2023 г. Общее число обработанных КТ-исследований ГМ составило 191 928. Каждый месяц для экспертной оценки случайным образом отбирали 80 КТ-исследований с балансом классов 70:30 (с предполагаемой патологией и нормой — 70 и 30% соответственно по оценке ИИ), согласно разработанной методологии [18].
Два врача-рентгенолога, специализирующиеся на нейровизуализации, со стажем более 3 лет оценивали эти КТ-исследования по двум главным критериям:
- соответствие трактовки (заключения);
- соответствие локализации (маркировки) патологической зоны.
Каждый из критериев мог иметь четыре вариации ответов:
- полное соответствие;
- частично корректная оценка;
- ложноположительные результаты — когда сервис обнаружил кровоизлияние там, где его нет;
- ложноотрицательные результаты — когда сервис не обнаружил кровоизлияние при его наличии.
Этическая экспертиза
Дизайн Эксперимента одобрен независимым этическим комитетом Московского рентгенологического общества (выписка из протокола № 2 НЭК МРО РОРР от 20.02.2020), также зарегистрирован на ClinicalTrials (NCT04489992).
Статистическая обработка
Для обработки полученных данных использовали метод построения и анализа характеристической кривой (ROC-анализ) посредством специально разработанного Web-инструмента5. Согласно результатам эмпирического исследования, минимальный объём набора данных для тестирования ИИ-сервиса в условиях периодического мониторинга должен составлять 400 КТ-исследований, при этом доля патологических случаев — не менее 10% [19]. Однако фактический объём набора данных оказался больше и составил 1200 КТ-исследований ГМ, при доле патологии 48,3%, что соответствовало нашим задачам. Для ИИ-сервиса определены метрики диагностической точности: чувствительность, специфичность, точность, AUROC. Ввиду наличия бинарного ответа от сервиса AUROC рассчитывали с учётом полученных значений чувствительности и специфичности. При расчёте метрик за ложноположительный принимали ответ сервиса о наличии ВЧК в исследовании при отсутствии данной патологии по мнению врача-эксперта, за ложноотрицательный — ответ сервиса об отсутствии ВЧК в исследовании при его наличии по мнению врача-эксперта. Общие метрики ИИ-сервиса, представленные в результатах, рассчитаны с 95% доверительным интервалом (ДИ) методом биномиального теста, поскольку исследуемые выборки содержат бинаризованные значения. Для оценки наличия и характера взаимосвязи между значением диагностических метрик и временем работы сервиса использовали коэффициент корреляции Пирсона (r). С целью сравнения значений диагностических метрик между калибровочными тестированиями применяли тест Манна–Уитни. При сопоставлении использовали одностороннюю версию теста с альтернативной гипотезой «медианное значение метрики до третьего калибровочного тестирования меньше, чем после него». Мы ожидали статистически значимый прирост метрических показателей после третьего калибровочного тестирования. Уровень значимости принятия статистических гипотез был равным 0,05.
РЕЗУЛЬТАТЫ
Калибровочное тестирование
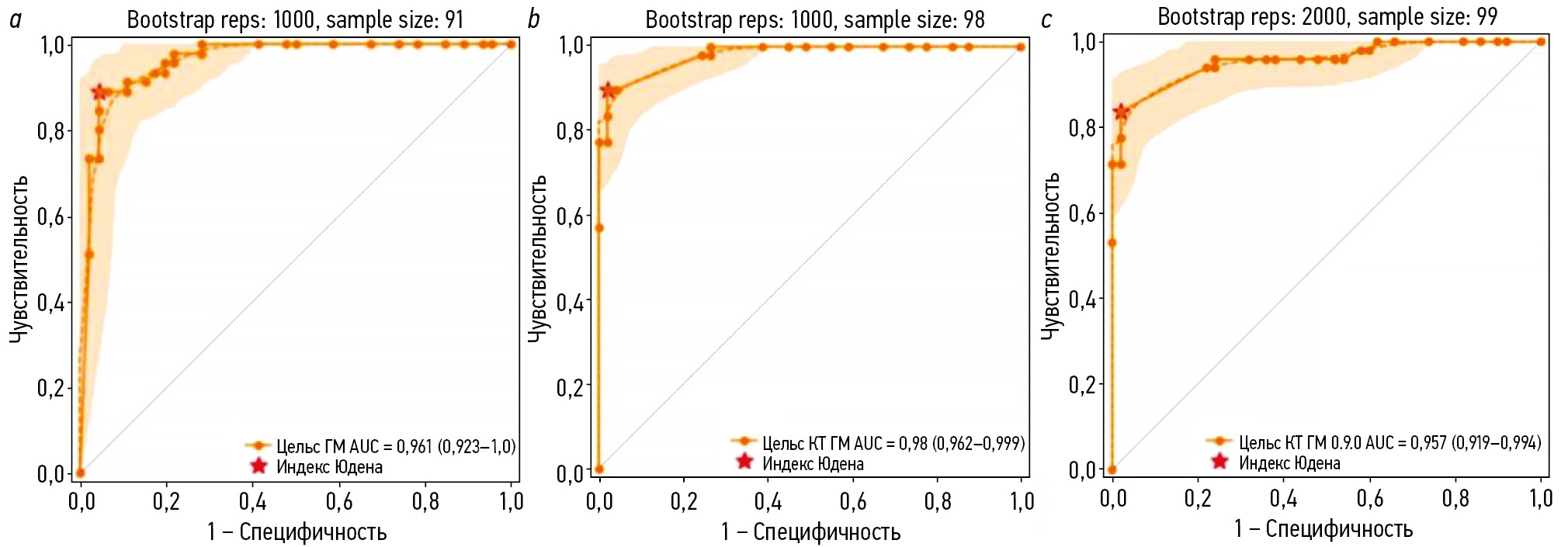
Количество КТ-исследований, не прошедших обработку по причине технической ошибки «сбой отправки исследования на тестирование» составило 9, 2 и 1 на первом, втором и третьем этапах калибровочного тестирования соответственно. По результатам анализа корректно обработанных КТ-исследований ГМ построены характеристические кривые, представленные на рис. 1.
Рис. 1. Характеристические кривые калибровочных тестирований сервиса искусственного интеллекта, предназначенного для автоматического анализа медицинских компьютерно-томографических изображений головного мозга на наличие внутричерепных кровоизлияний: a — первое; b — второе; с — третье.
В табл. 1 представлены численные метрики, которые мы получили при первом, втором и третьем калибровочных тестированиях.
Таблица 1. Метрики эффективности сервиса искусственного интеллекта, предназначенного для автоматического анализа медицинских компьютерно-томографических изображений головного мозга на наличие внутричерепных кровоизлияний, полученные по результатам трёх калибровочных тестирований
Параметры | Калибровка 1 0.6.11 | Калибровка 2 0.7.11 | Калибровка 3 0.8.31 |
AUROC, % (95% ДИ) | 96 (92–100) | 98 (96–99) | 96 (91–99) |
Чувствительность, % (95% ДИ) | 89 (80–98) | 90 (81–98) | 84 (73–94) |
Специфичность, % (95% ДИ) | 96 (90–100) | 98 (94–100) | 98 (94–100) |
Точность, % (95% ДИ) | 92 (87–98) | 94 (89–99) | 91 (85–97) |
Удельный вес ложноотрицательных результатов, % | 11 | 10 | 16 |
Удельный вес ложноположительных результатов, % | 4 | 2 | 2 |
Время обработки, с | 73 | 73 | 85 |
Примечание. Калибровка 1, 2, 3 соответствует первому, второму и третьему калибровочным тестированиям, проведённым на 0-, 3- и 7-м месяцах работы; 1 — версия сервиса искусственного интеллекта; AUROC — площадь под характеристической кривой. | |||
Необходимость их повтора была обусловлена доработками программного обеспечения, при котором его метрики эффективности могли ухудшиться. Калибровочные тестирования проводили после каждой доработки, которая сопровождалась изменениями ядра ИИ-сервиса. Однако во всех трёх случаях отличия в метриках эффективности были статистически незначимыми (p >0,05).
Клинический мониторинг
Результаты клинического мониторинга представлены в табл. 2.
Таблица 2. Матрица ошибок сервиса искусственного интеллекта и метрики эффективности по каждому месяцу
Месяц | Se, % | Sp, % | Ac, % | AUROC, % | ИП, n | ИО, n | ЛП, n | ЛО, n |
Калибровка 1 | ||||||||
1 | 100 | 39,4 | 50,0 | 93,1 | 14 | 26 | 40 | 0 |
2 | 100 | 46,0 | 57,5 | 94,7 | 17 | 29 | 34 | 0 |
3 | 100 | 42,2 | 53,7 | 99,4 | 16 | 27 | 37 | 0 |
Калибровка 2 | ||||||||
4 | 97,5 | 82,5 | 90,0 | 94,6 | 39 | 33 | 7 | 1 |
5 | 93,5 | 61,2 | 73,8 | 86,2 | 29 | 30 | 19 | 2 |
6 | 94,6 | 72,1 | 82,5 | 90,2 | 35 | 31 | 12 | 2 |
7 | 97,1 | 66,7 | 80,0 | 90,0 | 34 | 30 | 15 | 1 |
Калибровка 3 | ||||||||
8 | 100 | 75,6 | 87,5 | 93,9 | 39 | 31 | 10 | 0 |
9 | 95,3 | 79,5 | 87,8 | 92,8 | 41 | 31 | 8 | 2 |
10 | 100 | 71,1 | 83,8 | 92,8 | 35 | 32 | 13 | 0 |
11 | 100 | 68,9 | 82,5 | 92,2 | 35 | 31 | 14 | 0 |
12 | 97,6 | 76,9 | 87,5 | 93,1 | 40 | 30 | 9 | 1 |
13 | 100 | 78,0 | 88,8 | 94,5 | 39 | 32 | 9 | 0 |
14 | 97,6 | 73,7 | 86,3 | 92,2 | 41 | 28 | 10 | 1 |
15 | 97,4 | 83,3 | 90,0 | 94,7 | 37 | 35 | 7 | 1 |
16 | 95,6 | 82,9 | 90,0 | 93,8 | 43 | 29 | 6 | 2 |
17 | 94,4 | 77,3 | 85,0 | 91,7 | 34 | 34 | 10 | 2 |
18 | 100 | 88,9 | 95,0 | 97,2 | 44 | 32 | 4 | 0 |
Примечание. Калибровка 1, 2, 3 соответствует первому, второму и третьему калибровочным тестированиям. Серым цветом обозначены данные, исключённые из дальнейшего анализа. Se — чувствительность; Sp — специфичность; Ac — точность; AUROC — площадь под характеристической кривой; ИП — истинно положительный ответ; ИО — истинно отрицательный ответ; ЛП — ложноположительный ответ; ЛО — ложноотрицательный ответ. | ||||||||
Из представленных данных следует, что ИИ-сервис стабилизировал свои показатели после третьего месяца. Первые 3 мес. (апрель-июнь 2022 г.) были периодом апробации, в ходе которого разработчик дорабатывал своё решение — обработка исследований велась нестабильно, присутствовало большое количество технических ошибок, что влияло на результаты работы. Именно поэтому мы решили не включать данный период в дальнейший анализ.
Общая схема проведения клинического мониторинга ИИ-сервиса представлена на рис. 2.
Рис. 2. Схема анализа компьютерно-томографических исследований головного мозга при экспертной оценке в условиях клинического мониторинга: ВЧК+ — наличие внутричерепных кровоизлияний; ВЧК− — отсутствие внутричерепных кровоизлияний; ИП — истинно положительный результата; ЛП — ложноположительный результат; ЛО — ложноотрицательный результат; ИО — истинно отрицательный результат.
С момента стабилизации его работы суммарно проанализировано 1200 нативных изображений КТ ГМ (см. рис. 2). Средний возраст пациентов составил 61,2±18,6 года, из них 39% женщин. По результатам экспертной оценки (Ground Truth) признаки ВЧК содержали 580 КТ-исследований (48,3%). Результаты расчёта диагностических метрик за весь период с 3 по 18 мес. включительно представлены на рис. 3. По результатам бинарной классификации, проведённой ИИ-сервисом для выявления ВЧК, получены следующие значения метрик эффективности:
- чувствительность — 97,4% (95,8–98,5);
- специфичность — 75,4% (71,8–78,7);
- точность — 86,0% (83,9–87,9);
- AUROC — 94% (92,6–95,3).
Рис. 3. Динамика диагностических метрик работы сервиса искусственного интеллекта относительно результатов двух калибровочных тестирований: ось абсцисс — значения метрик; ось ординат — месяцы. Пунктиром обозначены результаты метрик, полученные в ходе калибровочных тестирований.
Корреляционный анализа выявил статистически значимую умеренную положительную корреляционную связь между временем работы и значениями диагностических метрик — специфичность и точность (для обеих метрик: r=0,5; p=0,04), AUROC (r=0,6; p=0,03). Для чувствительности статистически значимой взаимосвязи между её значением и временем работы ИИ-сервиса не выявлено.
Сравнение значений диагностических метрик между вторым и третьим калибровочными тестированиями, а также последующим этапом показало статистически значимый прирост только для чувствительности и специфичности (p=0,04).
Кроме того, мы оценили уточнённые диагностические метрики. Для их расчёта дополнительно проверено наличие не только факта обнаружения патологии, но и характер её локализации, а также соответствие описания врача ответу сервиса. В этом случае истинно положительным считали любой ответ, в котором присутствовало совпадение и по локализации, и по описанию детектированной патологии. Полное соответствие разметки и описания при наличии ВЧК достигнуто в 28,5% случаях (162 КТ-исследования). Соответственно, различия между разметкой и описанием отмечены в 71,5% (404 КТ-исследования). Результаты для уточнённых метрик эффективности следующие:
- чувствительность — 26,6% (22,9–30,4);
- специфичность — 73,8% (70,0–77,4);
- точность — 50,1% (47,1–53,0);
- AUROC — 49,6% (44,1–55,1).
Неточности в описании обнаружены в 61 случае — ИИ-сервис корректно идентифицировал наличие патологии на снимках, но либо пропускал отдельные очаги кровоизлияний при их множественности, либо ошибочно классифицировал его тип. Неточности разметки зафиксированы в 64 КТ-исследованиях, что включало некорректную сегментацию участков кровоизлияний. Однако наиболее часто неточность наблюдали одновременно и в описаниях, и в разметке (279 КТ-исследований) (см. рис. 2).
Одним из наиболее частых примеров частично корректного срабатывания можно назвать случаи, когда при множественных кровоизлияниях ИИ-сервис выделял один тип кровоизлияния, но пропускал другой. Так, на рис. 4, a слева пропущено внутримозговое кровоизлияние в левом полушарии, а на рис. 4, c слева ИИ-сервис корректно его сегментировал в левом полушарии, но не отметил внутрижелудочковое кровоизлияние в правом полушарии, а также межполушарное субарахноидальное кровоизлияние и по бороздам в обоих полушариях. Также частым случаем некорректных срабатываний является частичная сегментация кровоизлияния с ошибочным определением его типа (см. рис. 4, c) — справа представлено субдуральное кровоизлияние, которое ИИ-сервис идентифицировал как субарахноидальное. Гораздо реже среди примеров частично корректного срабатывания можно отметить полностью корректную сегментацию патологической зоны кровоизлияния, но неверное определение его/их типа (см. рис. 4, b) — справа внутримозговое кровоизлияние ошибочно классифицировано как субдуральное, а слева субдуральное кровоизлияние отнесено к субарахноидальному.
Рис. 4. Примеры частично корректных срабатываний сервиса искусственного интеллекта: a — корректное определение типа кровоизлияния и некорректная их сегментация; b — корректная сегментация областей кровоизлияния, ошибочное определение их типа; c — частичное выделение одних кровоизлияний и пропуск других, некорректные как сегментация, так и определение типа.
Всего отмечено 152 ложноположительных результата. Наиболее частыми их причинами являлись сегментации магистральных артерий, венозных синусов и частично кальцифицированных оболочек мозга (рис. 5, a).
Рис. 5. Примеры ложно-положительных (a) и ложноотрицательных (b) срабатываний сервиса искусственного интеллекта.
ИИ-сервис не выявил патологические изменения на 14 изображениях КТ ГМ. Наиболее часто встречали ложноотрицательные результаты при субарахноидальных кровоизлияниях — 8 случаев (см. рис. 5, b, в центре и справа). Реже наблюдали пропуски внутрижелудочковых кровоизлияний — 2 случая (см. рис. 5, b, слева), внутримозговых кровоизлияний — 2 случая, а также по одному случаю субдурального и эпидурального кровоизлияния.
ОБСУЖДЕНИЕ
Проведённое исследование не является первым среди клинической апробации ИИ-сервисов для выявления ВЧК по данным бесконтрастной КТ ГМ. Однако его отличительной особенностью является длительность (18 мес.) и многоцентровой характер (56 стационаров, 248 врачей-рентгенологов, составивших первичное заключение).
В нашем исследовании в рамках клинического мониторинга отмечен статистически значимый прирост медианных значений для двух из четырёх основных диагностических метрик. Причём следует отметить различную величину прироста. Изначально довольно высокие показатели чувствительности (Me 95,8%) со временем наблюдения статистически значимо (p=0,04) повысились (Me 97,4%). Для специфичности были характерны сравнительно низкие начальные значения (Me 69,4%) и статистически значимый (p=0,04) прирост (Me 76,2%). Для точности и AUROC статистически значимых изменений не наблюдали (p=0,1).
Стоит отметить различия в значениях метрик эффективности, полученных при калибровочных тестированиях и клиническом мониторинге. Их расчёт при калибровочных тестированиях проводили с использованием индекса Юдена при установленном оптимальном пороге в 75%. Выявлена статистически значимая положительная корреляция между временем работы сервиса и специфичностью, точностью, AUROC (r от 0,5 до 0,6), при этом статистически значимый прирост значения зафиксирован только для специфичности и чувствительности.
ВЧК — неотложная патология, в большинстве случаев требующая быстрого реагирования и повышающая риски ухудшения состояния пациентов. Именно поэтому приоритетной задачей является настройка ИИ-сервиса на максимальную чувствительность к наличию патологии, что позволяет в первую очередь привлекать внимание врача к пациентам с подозрением на ВЧК. Однако автоматическая подстройка порога для оптимизации всех диагностических параметров должна происходить при условии, что это не приводит к критическому снижению специфичности.
В нескольких одноцентровых исследованиях продемонстрирована высокая специфичность, варьирующая от 91 до 98%, однако чувствительность оказывалась ниже — в диапазоне от 81 до 94% [20–23].
В единичных исследованиях, проводимых на базе нескольких клинических центров и сопоставимых с настоящим по условиям и объёму, также отмечают более низкие значения чувствительности при высокой специфичности [24–27]. Так, J. McLouth и соавт. [24] при использовании коммерчески доступного сервиса ИИ CINA® v1.0 (Avicenna.ai, Франция) сообщили о чувствительности и специфичности — 91,4 и 97,5% соответственно, полученных на выборке 814 пациентов с долей патологии 31%. Похожие показатели эффективности продемонстрированы в работе A. Kundisch и соавт. [25], где при использовании коммерческого программного обеспечения для выявления ВЧК (AIDOC, Израиль) получены значения чувствительности и специфичности — 87,6 и 92,8% соответственно. Исследование провели на выборке из 4946 пациентов с долей патологии 5%. Два крупных недавних исследования — A.J. Del Gaizo и соавт. [26] с выборкой 58 321 (доля патологии 2,7%) и G. Pettet и соавт. [27] с выборкой 1315 (доля патологии 8,5%) — проведённые также с применением коммерческих сервисов ИИ [CINA® v1.0 (Avicenna.ai, Франция) и qER® v2.0 (Qure.ai, Индия)], предоставили следующие значения чувствительности и специфичности: 75,6 и 92,1; 85,7 и 94,3% соответственно.
По данным систематического обзора S.M. Mäenpää и соавт. [28], посвящённого оценке диагностической точности моделей ИИ для экстренного анализа результатов КТ ГМ в условиях внешней клинической апробации, установлено, что большинство коммерчески доступных сервисов ИИ имели, как правило, более низкую чувствительность и положительную прогностическую значимость, которая отражает количество ложноположительных ответов. Это свидетельствует о более слабой обобщаемости и не совсем подходит для сценария сортировки и приоритизации рабочего списка посредством пометки ВЧК-положительных сканов из-за более высокого риска «утомления бдительности». В нашем исследовании мы получили высокие показатели чувствительности, однако специфичность на уровне 75,4% обусловлена большим количеством ложноположительных результатов и указывает на вероятную дополнительную нагрузку на внимание врача. В некоторых исследованиях снижение прогностической значимости объясняли более низкой распространённостью целевого состояния, что отражает естественное влияние базовой частоты патологии на метрики [23, 29]. Приемлемый уровень ложноположительных ответов ИИ-сервиса на потоке требует дальнейшего изучения.
Об ограничениях в использовании всего функционала ИИ-сервиса (корректное определение типа кровоизлияний и их локализация) говорят и довольно низкие (за исключением специфичности) значения уточнённых метрик. Тем не менее нужно отметить, что представленные метрики рассчитаны исключительно для случаев, в которых ответ ИИ-сервиса полностью совпадал с мнением врача-эксперта как по разметке, так и по типу кровоизлияния. Общие же показатели метрик эффективности достаточно высокие, в частности, чувствительность превышает значения, описанные в литературе.
Некоторые авторы отмечают превосходную согласованность разметки объёмов патологических областей плотности вследствие ВЧК в ГМ между коммерчески доступным сервисом ИИ и полуавтоматической разметкой. Однако N. Schmitt и соавт. [30] отмечают, что при чувствительности и специфичности 91 и 89% соответственно (на выборке с долей патологии 50%) ИИ может служить «вторым мнением» для врача, но не подходит для самостоятельного использования, с чем согласны и другие авторы. Наши данные демонстрируют, что полное совпадение описаний и разметки патологических областей, выполненных исследуемым ИИ-сервисом, с заключениями врачей невозможно достигнуть даже в 1/3 всех случаев с патологией, что в значительной степени связано с сочетанием различных типов кровоизлияний. Именно поэтому ИИ-сервис можно применять как вспомогательный инструмент для первичного выявления патологии, однако пока он не будет существенно доработан в части качества разметки и классификации кровоизлияний, нецелесообразно его использование в виде полноценной системы поддержки принятия врачебных решений, предназначенной для углублённой диагностики типов кровоизлияний и их объёмов.
Постоянная доработка ИИ-сервисов и адаптация к изменяющимся клиническим условиям необходима и технически возможна [31]. Использование в качестве основы глубоких свёрточных нейронных сетей позволяет этому программному обеспечению эффективнее извлекать и анализировать сложные признаки изображений, которые недоступны глазу человека, сопоставляя их в контексте отличной от человеческой логики, что действительно способно приводить к повышению точности диагностики [26, 32]. Обучение на новых данных способствует улучшению производительности с течением времени [33], и в нашем исследовании с периодом наблюдения более года это продемонстрировано. Кроме того, необходимо обеспечить регулярный — желательно ежемесячный — контроль производительности ИИ-сервиса на независимых клинических данных [21], а также организовать систематическую обратную связь от врачей. Это позволит разработчикам определить необходимость в дополнительных обучающих данных и откорректировать пороговые значения, оптимизируя баланс между чувствительностью и точностью [32].
Существует ещё одна важная причина, по которой необходим непрерывный клинический мониторинг качества работы ИИ-сервиса. Наши данные показывают, что он обеспечивает более объективную оценку, чем тестирование в лабораторных условиях, даже при использовании внешних валидационных данных (в нашем случае — калибровочные тестирования). Кроме того, относительно низкий показатель специфичности должен побуждать врачей к осторожному использованию ИИ-сервиса в реальной клинической практике без должного контроля, поскольку это может привести к увеличению числа необоснованных госпитализаций или даже к ненужным хирургическим вмешательствам [34]. Одновременно с этим высокая чувствительность даёт возможность с высокой точностью исключать кровотечения в случае острого ишемического инсульта, что определяет пригодность пациента для проведения тромболитической терапии [27]. Таким образом, оптимальное использование ИИ рентгенологами может быть дополнено пониманием сценариев, в которых он, вероятно, будет генерировать неточные выходные данные. Кроме того, подсчёт объёмов кровоизлияний с помощью ИИ-сервиса (при наличии соответствующего функционала) может служить эффективным способом объективизации корректности разметки. Данную задачу необходимо рассматривать в рамках дальнейших исследований.
Ограничения исследования
Наше исследование имеет несколько ограничений. Во-первых, мы не анализировали диагностические метрики ИИ-сервиса по каждому типу кровоизлияний и точность площади маркировки патологических областей. Наша концепция обсервационного ретроспективного многоцентрового анализа направлена на выявление изменений в его производительности во времени на основе фактической клинической работы. Во-вторых, наша выборка для клинического мониторинга была насыщена ВЧК (~50%) и не соответствовала реальной распространённости патологии в популяции (~8–12%), что могло способствовать увеличению количества ложноположительных результатов и снижению специфичности по сравнению с теми метриками эффективности, которые заявлены производителем. Это подчёркивает необходимость стандартизации клинически ориентированного обучения и проверки ИИ в соответствующих условиях. Тем не менее сохраняющаяся высокая чувствительность ИИ-сервиса, даже при увеличении доли случаев с патологией в выборке, свидетельствует о его высокой способности выявлять критически значимые патологические изменения, что в аспекте неотложной медицинской помощи следует рассматривать как преимущество.
ЗАКЛЮЧЕНИЕ
В ходе 18-месячного ретроспективного наблюдения за работой ИИ-сервиса для обнаружения ВЧК по данным неконтрастной КТ ГМ в 56 стационарах г. Москвы удалось продемонстрировать многообещающие результаты с очень высокой чувствительностью (97,4%) и разумной специфичностью (75,4%), которые с течением времени улучшались. Однако низкие значения уточнённых метрик (чувствительность и точность — 26,6 и 50,1% соответственно) указывают на значительные расхождения между оценками рентгенологов и ИИ-сервиса, связанные с неполной сегментацией патологических зон и ошибками в классификации типов ВЧК. Рентгенологам необходимо понимать особенности работы ИИ в клинической практике и учитывать, что положительный результат не всегда свидетельствует о наличии кровоизлияния, а выявленное кровоизлияние может быть не единственным и не всегда точно сегментировано. Разработчикам подобного программного обеспечения необходимо сосредоточиться на снижении количества ложноположительных ответов и повышении качества работы ИИ-сервиса, чтобы его функции были клинически полезными. Тем не менее текущая конфигурация позволяет исключать кровоизлияние с очень высокой вероятностью, что особенно полезно для неотложной сортировки пациентов в приёмных отделениях.
ДОПОЛНИТЕЛЬНАЯ ИНФОРМАЦИЯ
Вклад авторов. А.Н. Хоружая — сбор и анализ литературных данных, тестирование сервиса искусственного интеллекта, анализ данных мониторинга, написание и редактирование текста рукописи; К.М. Арзамасов — концепция исследования, организация тестирования сервиса искусственного интеллекта, сбор данных для мониторинга, редактирование текста рукописи; М.Р. Коденко — статистическая обработка данных, написание и редактирование текста рукописи; Е.И. Кремнёва — сбор и анализ литературных данных, анализ данных мониторинга, редактирование текста рукописи; Д.В. Буренчев — концепция исследования, анализ литературных данных, редактирование текста рукописи. Все авторы одобрили рукопись (версию для публикации), а также согласились нести ответственность за все аспекты работы, гарантируя надлежащее рассмотрение и решение вопросов, связанных с точностью и добросовестностью любой её части.
Благодарности. Авторы выражают благодарность главному научному сотруднику Научно-практического клинического центра диагностики и телемедицинских технологи, д.м.н. А.В. Петряйкину за помощь в проведении исследования.
Этическая экспертиза. Дизайн Эксперимента одобрен независимым этическим комитетом Московского рентгенологического общества (выписка из протокола № 2 НЭК МРО РОРР от 20.02.2020), также зарегистрирован на ClinicalTrials (NCT04489992).
Источники финансирования. Данная статья подготовлена авторским коллективом в рамках научно-исследовательской работы «Научные методологии устойчивого развития технологий искусственного интеллекта в медицинской диагностике», (ЕГИСУ: № 123031500004-5).
Раскрытие интересов. Авторы заявляют об отсутствии отношений, деятельности и интересов за последние три года, связанных с третьими лицами (коммерческими и некоммерческими), интересы которых могут быть затронуты содержанием статьи.
Оригинальность. При создании настоящей работы авторы не использовали ранее опубликованные сведения (текст, иллюстрации, данные).
Доступ к данным. Редакционная политика в отношении совместного использования данных к настоящей работе не применима.
Генеративный искусственный интеллект. При создании настоящей статьи технологии генеративного искусственного интеллекта не использовали.
Рассмотрение и рецензирование. Настоящая работа подана в журнал в инициативном порядке и рассмотрена в соответствии с процедурой fast-track. В рецензировании участвовали три внешних рецензента и научный редактор издания.
ADDITIONAL INFORMATION
Author contributions: A.N. Khoruzhaya: published data search and analysis, AI service testing, monitoring data analysis, writing—original draft, writing—review & editing; K.M. Arzamasov: study conceptualization, organization of AI service testing, monitoring data collection, writing—review & editing; M.R. Kodenko: formal analysis, writing—original draft, writing—review & editing; E.I. Kremneva: published data search and analysis, monitoring data analysis, writing—review & editing; D.V. Burenchev: study conceptualization, published data analysis, writing—review & editing. All the authors approved the version of the manuscript to be published and agreed to be accountable for all aspects of the work, ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Acknowledgments: The authors express their gratitude to Prof. A.V. Petryaykin, Dr. Sci. (Medicine), Chief Researcher at the Scientific and Practical Clinical Center for Diagnostics and Telemedicine Technologies, for his assistance in the study.
Ethics approval: The study design was approved by the Independent Ethics Committee of the Moscow Radiological Society (Extract from Protocol No. 2 of the IEC of MRO RSRR dated February 20, 2020) and registered on ClinicalTrials (NCT04489992).
Funding sources: This article was part of the research project Scientific Methods for the Sustainable Development of Artificial Intelligence Technologies in Medical Diagnostics (Unified State Information Accounting System No. 123031500004-5).
Disclosure of interests: The authors have no relationships, activities, or interests for the last three years related to for-profit or not-for-profit third parties whose interests may be affected by the content of the article.
Statement of originality: No previously published material (text, images, or data) was used in this work.
Data availability statement: The editorial policy regarding data sharing does not apply to this work.
Generative AI: No generative artificial intelligence technologies were used to prepare this article.
Provenance and peer review: This paper was submitted unsolicited and reviewed following the fast-track procedure. The peer review process involved three external reviewers and the in-house science editor.
1 Технологии искусственного интеллекта в здравоохранении. В: Центр диагностики и телемедицины [интернет]. Москва: Центр диагностики и телемедицины, 2020–2024. Режим доступа: https://mosmed.ai/ Дата обращения: 13.12.2024.
2 Базовые диагностические требования к результатам работы ИИ-сервисов [интернет]. В: Научно-практический клинический центр диагностики и телемедицинских технологи, 2024–2024. Режим доступа: https://mosmed.ai/ai/docs/ Дата обращения: 13.12.2024.
3 Базовые функциональные требования к результатам работы ИИ-сервисов [интернет]. В: Научно-практический клинический центр диагностики и телемедицинских технологи, 2024–2024. Режим доступа: https://mosmed.ai/ai/docs/ Дата обращения: 13.12.2024.
4 Свидетельство о государственной регистрации базы данных № 2022620559/ 16.03.2022. Бюл. № 3. Морозов С.П., Павлов Н.А., Петряйкин А.В., и др. MosMedData: набор диагностических компьютерно-томографических изображений головного мозга с наличием и отсутствием признаков внутричерепного кровоизлияния. Режим доступа: https://www.elibrary.ru/item.asp?id=48137428 Дата обращения: 13.12.2024.
5 Инструмент для ROC-анализа диагностических тестов [интернет]. В: Научно-практический клинический центр диагностики и телемедицинских технологи, 2022–2024. Режим доступа: https://roc-analysis.mosmed.ai/ Дата обращения: 13.12.2024.
About the authors
Anna N. Khoruzhaya
Research and Practical Clinical Center for Diagnostics and Telemedicine Technologies
Author for correspondence.
Email: KhoruzhayaAN@zdrav.mos.ru
ORCID iD: 0000-0003-4857-5404
SPIN-code: 7948-6427
MD
Russian Federation, MoscowKirill M. Arzamasov
Research and Practical Clinical Center for Diagnostics and Telemedicine Technologies
Email: ArzamasovK@zdrav.mos.ru
ORCID iD: 0000-0001-7786-0349
SPIN-code: 3160-8062
MD, Dr. Sci. (Medicine)
Russian Federation, MoscowMaria R. Kodenko
Research and Practical Clinical Center for Diagnostics and Telemedicine Technologies
Email: KodenkoM@zdrav.mos.ru
ORCID iD: 0000-0002-0166-3768
SPIN-code: 5789-0319
Cand. Sci. (Engineering)
Russian Federation, MoscowElena I. Kremneva
Research and Practical Clinical Center for Diagnostics and Telemedicine Technologies; Russian Center of Neurology and Neurosciences
Email: KremnevaE@zdrav.mos.ru
ORCID iD: 0000-0001-9396-6063
SPIN-code: 8799-8092
MD, Dr. Sci. (Medicine)
Russian Federation, Moscow; MoscowDmitry V. Burenchev
Research and Practical Clinical Center for Diagnostics and Telemedicine Technologies
Email: BurenchevD@zdrav.mos.ru
ORCID iD: 0000-0003-2894-6255
SPIN-code: 2411-3959
MD, Dr. Sci. (Medicine)
Russian Federation, MoscowReferences
- Li X, Zhang L, Wolfe CDA, Wang Y. Incidence and long-term survival of spontaneous intracerebral hemorrhage over time: a systematic review and meta-analysis. Frontiers in Neurology. 2022;13:819737. doi: 10.3389/fneur.2022.819737 EDN: MLOQRJ
- Hemorrhagic stroke: clinical guidelines. Moscow: Ministry of Health of the Russian Federation; 2022. (In Russ.) [cited 2024 Dec 12]. Available from: https://ruans.org/Text/Guidelines/hemorrhagic-stroke-2022.pdf
- Hostettler IC, Seiffge DJ, Werring DJ. Intracerebral hemorrhage: an update on diagnosis and treatment. Expert Review of Neurotherapeutics. 2019;19(7):679–694. doi: 10.1080/14737175.2019.1623671 EDN: JWSYUZ
- Woo D, Comeau ME, Venema SU, et al. Risk factors associated with mortality and neurologic disability after intracerebral hemorrhage in a racially and ethnically diverse cohort. JAMA Network Open. 2022;5(3):e221103. doi: 10.1001/jamanetworkopen.2022.1103 EDN: BVHNLU
- Yaghi S, Dibu J, Achi E, et al. Hematoma expansion in spontaneous intracerebral hemorrhage: predictors and outcome. International Journal of Neuroscience. 2014;124(12):890–893. doi: 10.3109/00207454.2014.887716
- Gong B, Khalvati F, Ertl-Wagner BB, Patlas MN. Artificial intelligence in emergency neuroradiology: current applications and perspectives. Diagnostic and Interventional Imaging. 2025;106(4):135–142. doi: 10.1016/j.diii.2024.11.002 EDN: DHXSGS
- Arbabshirani MR, Fornwalt BK, Mongelluzzo GJ, et al. Advanced machine learning in action: identification of intracranial hemorrhage on computed tomography scans of the head with clinical workflow integration. npj Digital Medicine. 2018;1(1):9. doi: 10.1038/s41746-017-0015-z EDN: BORIWC
- Seyam M, Weikert T, Sauter A, et al. Utilization of artificial intelligence–based intracranial hemorrhage detection on emergent noncontrast CT images in clinical workflow. Radiology: Artificial Intelligence. 2022;4(2):e210168. doi: 10.1148/ryai.210168 EDN: HEPSBX
- Davis MA, Rao B, Cedeno PA, et al. machine learning and improved quality metrics in acute intracranial hemorrhage by noncontrast computed tomography. Current Problems in Diagnostic Radiology. 2022;51(4):556–561. doi: 10.1067/j.cpradiol.2020.10.007 EDN: NHQFYC
- O’Neill TJ, Xi Y, Stehel E, et al. Active reprioritization of the reading worklist using artificial intelligence has a beneficial effect on the turnaround time for interpretation of head CT with intracranial hemorrhage. Radiology: Artificial Intelligence. 2021;3(2):e200024. doi: 10.1148/ryai.2020200024 EDN: LCDGTM
- Smorchkova AK, Khoruzhaya AN, Kremneva EI, Petryaikin AV. Machine learning technologies in CT-based diagnostics and classification of intracranial hemorrhages. Burdenko's Journal of Neurosurgery. 2023;87(2):85. doi: 10.17116/neiro20238702185EDN: JVZDST
- Yu KH, Kohane IS. Framing the challenges of artificial intelligence in medicine. BMJ Quality & Safety. 2018;28(3):238–241. doi: 10.1136/bmjqs-2018-008551
- Allen B, Dreyer K, Stibolt R, et al. Evaluation and real-world performance monitoring of artificial intelligence models in clinical practice: try it, buy it, check it. Journal of the American College of Radiology. 2021;18(11):1489–1496. doi: 10.1016/j.jacr.2021.08.022 EDN: NMKGVD
- Recht MP, Dewey M, Dreyer K, et al. Integrating artificial intelligence into the clinical practice of radiology: challenges and recommendations. European Radiology. 2020;30(6):3576–3584. doi: 10.1007/s00330-020-06672-5 EDN: WWDEXB
- Vasiliev YuA, Vlazimirskyy AV, Omelyanskaya OV, et al. Methodology for testing and monitoring artificial intelligence-based software for medical diagnostics. Digital Diagnostics. 2023;4(3):252–267. doi: 10.17816/DD321971 EDN: UEDORU
- Morozov SP, Vladzimirsky AV, Klyashtornyy VG, et al. Clinical acceptance of software based on artificial intelligence technologies (radiology). Moscow: Research and Practical Clinical Center for Diagnostics and Telemedicine Technologies; 2019. EDN: GWJIMI
- Morozov SP, Vladzimirsky AV, Andreychenko AE, et al. Regulations for the preparation of data sets with a description of approaches to the formation of a representative data sample. Moscow: Research and Practical Clinical Center for Diagnostics and Telemedicine Technologies; 2022. (In Russ.) EDN: XENAJE
- Chetverikov SF, Arzamasov KM, Andreichenko AE, et al. Approaches to sampling for quality control of artificial intelligence in biomedical research. Sovremennye tehnologii v medicine. 2023;15(2):19. doi: 10.17691/stm2023.15.2.02 EDN: FUKXYC
- Kodenko MR, Bobrovskaya TM, Reshetnikov RV, et al. Empirical approach to sample size estimation for testing of AI algorithms. Doklady Mathematics. 2024;110(S1):S62–S74. doi: 10.1134/S1064562424602063 EDN: VJHJRD
- Salehinejad H, Kitamura J, Ditkofsky N, et al. A real-world demonstration of machine learning generalizability in the detection of intracranial hemorrhage on head computerized tomography. Scientific Reports. 2021;11(1):17051. doi: 10.1038/s41598-021-95533-2 EDN: SXLMCH
- Zia A, Fletcher C, Bigwood S, et al. Retrospective analysis and prospective validation of an AI-based software for intracranial haemorrhage detection at a high-volume trauma centre. Scientific Reports. 2022;12(1):19885. doi: 10.1038/s41598-022-24504-y EDN: IWNBET
- Ginat DT. Analysis of head CT scans flagged by deep learning software for acute intracranial hemorrhage. Neuroradiology. 2019;62(3):335–340. doi: 10.1007/s00234-019-02330-w EDN: WTOITQ
- Voter AF, Meram E, Garrett JW, Yu JPJ. Diagnostic accuracy and failure mode analysis of a deep learning algorithm for the detection of intracranial hemorrhage. Journal of the American College of Radiology. 2021;18(8):1143–1152. doi: 10.1016/j.jacr.2021.03.005 EDN: GPJYDS
- McLouth J, Elstrott S, Chaibi Y, et al. Validation of a deep learning tool in the detection of intracranial hemorrhage and large vessel occlusion. Frontiers in Neurology. 2021;12:656112. doi: 10.3389/fneur.2021.656112 EDN: FFIXVV
- Kundisch A, Hönning A, Mutze S, et al. Deep learning algorithm in detecting intracranial hemorrhages on emergency computed tomographies. PLOS ONE. 2021;16(11):e0260560. doi: 10.1371/journal.pone.0260560 EDN: QPACKZ
- Del Gaizo AJ, Osborne TF, Shahoumian T, Sherrier R. Deep learning to detect intracranial hemorrhage in a national teleradiology program and the impact on interpretation time. Radiology: Artificial Intelligence. 2024;6(5):e240067. doi: 10.1148/ryai.240067 EDN: EHHAOO
- Pettet G, West J, Robert D, et al. A retrospective audit of an artificial intelligence software for the detection of intracranial haemorrhage used by a teleradiology company in the United Kingdom. BJR|Open. 2023;6(1):tzae033. doi: 10.1093/bjro/tzae033 EDN: DWNYCF
- Mäenpää SM, Korja M. Diagnostic test accuracy of externally validated convolutional neural network (CNN) artificial intelligence (AI) models for emergency head CT scans – A systematic review. International Journal of Medical Informatics. 2024;189:105523. doi: 10.1016/j.ijmedinf.2024.105523 EDN: HLVVYQ
- Eldaya RW, Kansagra AP, Zei M, et al. Performance of automated RAPID intracranial hemorrhage detection in real-world practice: a single-institution experience. Journal of Computer Assisted Tomography. 2022;46(5):770–774. doi: 10.1097/rct.0000000000001335 EDN: GRDZTF
- Schmitt N, Mokli Y, Weyland CS, et al. Automated detection and segmentation of intracranial hemorrhage suspect hyperdensities in non-contrast-enhanced CT scans of acute stroke patients. European Radiology. 2021;32(4):2246–2254. doi: 10.1007/s00330-021-08352-4 EDN: OLFWXI
- Warman R, Warman A, Warman P, et al. Deep learning system boosts radiologist detection of intracranial hemorrhage. Cureus. 2022;undefined:. doi: 10.7759/cureus.30264 EDN: IRZKDY
- Buchlak QD, Tang CHM, Seah JCY, et al. Effects of a comprehensive brain computed tomography deep learning model on radiologist detection accuracy. European Radiology. 2023;34(2):810–822. doi: 10.1007/s00330-023-10074-8 EDN: ZHIFOG
- Ngiam KY, Khor IW. Big data and machine learning algorithms for health-care delivery. The Lancet Oncology. 2019;20(5):e262–e273. doi: 10.1016/S1470-2045(19)30149-4
- Kiefer J, Kopp M, Ruettinger T, et al. Diagnostic accuracy and performance analysis of a scanner-integrated artificial intelligence model for the detection of intracranial hemorrhages in a traumatology emergency department. Bioengineering. 2023;10(12):1362. doi: 10.3390/bioengineering10121362 EDN: EPLIBY
Supplementary files