Changing of pulmonary artery diameter in accordance with severity of COVID-19 (assessment based on non-contrast computer tomography)
- Authors: Aliev A.F.1,2, Kudryavtsev N.D.1,3, Petraikin A.V.3, Artyukova Z.R.3, Shkoda A.S.4, Morozov S.P.3
-
Affiliations:
- L.A. Vorokhobov Municipal Clinical Hospital No 67 of the Moscow Health Department
- The Moscow Research and Clinical Center for Tuberculosis Control of the Moscow Health Department
- Moscow Center for Diagnostics and Telemedicine
- Vorokhobov City Clinical Hospital No. 67
- Issue: Vol 2, No 3 (2021)
- Pages: 249-260
- Section: Original Study Articles
- Submitted: 26.07.2021
- Accepted: 07.09.2021
- Published: 15.10.2021
- URL: https://jdigitaldiagnostics.com/DD/article/view/76726
- DOI: https://doi.org/10.17816/DD76726
- ID: 76726
Cite item
Abstract
BACKGROUND: Computed tomography is the method of choice for assessing the volume of lung damage in viral pneumonia, including those associated with COVID-19. In addition, computed tomography can determine the main vessels size of the thorax. This allowed us to analyze the relationship between the severity of COVID-19 and the changes in the diameters of the pulmonary artery (PA) and ascending aorta (Ao). Dilation of the PA is a sign of pulmonary hypertension. The study of these patterns may be of clinical significance in determining the treatment tactics and prognosis of the course of COVID-19 disease.
AIM: To evaluate the relationship between PA diameter and the severity of the COVID-19 course in patients of different ages.
MATERIALS AND METHODS: This study is a single-centered, cross-section, continuous, uncontrolled study performed on a group of patients (n=511, 267 men, median 59 years, IQR 49.0–65.0, ages 31–84 years) who were treated in a temporary hospital to treat patient with COVID-19. During hospitalization all patients were examined by CT scan of the chest. All studies were carried out using a mobile CT scan system Airo TruCT (Stryker, USA). The degree of damage to the lung tissue was assessed using the CT volume scale 1–4. Measurement of the LA and Ao diameters was carried out using standard instruments of the radiologist’s CT workstation perpendicular to the long axis of the vessel.
RESULTS: The following statistically significant regularities were obtained: the detection of a dilated pulmonary artery (PA) and an increased PA/Ao ratio correlated to an increase in the degree of lung damage in COVID-19 (Kruskal-Wallis test, K-W p <0.001; median test, MT p <0.001), the diameter of the ascending aorta (Ao) significantly increases with the patient’s age (K-W p <0.001; MT p <0.001). An insignificant correlation between an increase in the diameter of the pulmonary artery (PA) and the patient’s age (K-W p=0.094; MT p=0.311) and an insignificant correlation between detection of a change in aortic (Ao) diameter and the degree of lung damage (K-W p=0.061; MT p=0.165) were shown. In groups with a severe course of the disease and a large volume of lung lesions (CT-3 and CT-4), a significantly greater number of patients with signs of pulmonary hypertension (detection of the dilated pulmonary artery: 29 mm and more) was shown for all age groups.
CONCLUSION: The study showed that PA dilatation and increased PA/Ao diameter ratio were significantly associated with increased pulmonary lesion volume in COVID-19 in all age groups.
Full Text
BACKGROUND
In the context of the coronavirus disease-2019 (COVID-19) pandemic, pulmonary computed tomography (CT) became one of the leading methods for COVID-19 severity diagnosis and assessment [1–3].
The federal clinical guidelines for the prevention, diagnosis, and treatment defined four degrees of COVID-19 severity [2]. This division is based on several clinical and laboratory parameters, including the volume of pulmonary tissue lesions according to grades 1–4 of the CT classification [1, 3]. Patients with a mild degree of disease severity had no lung lesions (CT–0); moderate disease severity had up to 25% (CT–1) and 50%–75% (CT–2) lung involvement degree; 50%–75% (CT–3) in severe disease course; and over 70% (CT–4) in extremely severe disease. In addition, the degree of pneumonia severity according to the CT classification has prognostic value. Thus, a significantly increased proportion of patients who died depending on the volume of the affected pulmonary parenchyma according to the CT scale was shown, including a jump in the risk of lethality when transitioning from mild and moderate to severe and extremely severe disease course (from CT 1–2 to CT 3–4) [4].
The radiological sign analyses, which are typical for COVID-19-associated pneumonia, paid considerable attention to the pulmonary parenchyma, pleura, and bronchial tree assessment, and to a lesser extent, to the great vessel changes [5, 6]. The most characteristic and stable signs of COVID-19-associated pneumonia are segmental pulmonary artery (PA) dilation [7] and high risks of pulmonary embolism and acute respiratory distress syndrome, which is accompanied by gas exchange worsening, hypoxemia, and multiple organ failure [4, 7, 8].
A significantly increased PA diameter and PA and ascending aorta (Asc Ao) diameter ratio is shown in patients with lethal outcomes compared to those who recovered [9].
Thus, the lung lesion volume and the state of the pulmonary vessels (both segmental PAs, [10–12] and the pulmonary trunk [main PA]) are considered to assess the course of COVID-19, current therapy effectiveness, and prognosis using CT [9]. Increased pressure in the arterial vascular bed of the pulmonary circulation indicates the difficulty of blood transit through the lungs, which reduces the patient’s compensatory abilities, leading to heart failure.
AIM: This study is aimed to determine the relationship between the COVID-19 severity and the PA and Asc Ao diameters in patients of different ages.
METHODS
Study design
A single-center, cross-sectional, continuous, and non-controlled study based on chest CT was conducted (Fig. 1).
Fig. 1. Study design
CT: computed tomography; CT 1–4: adapted scale for visual assessment of the relationship between the severity of the patient’s general condition and the nature and intensity of radiological signs of pulmonary changes in COVID-19; PA: pulmonary artery.
Eligibility criteria
Inclusion criteria are COVID-19 diagnosis that is confirmed by polymerase chain reaction, signs of viral pneumonia on CT scan, and voluntary informed consent for medical intervention. Chest CT scans were performed for the first time in a reserve hospital during admission or within 4 days after admission, provided that chest CT scans were performed in another medical institution at the prehospital stage [3].
Exclusion criteria are the history of chronic lung disease diagnosis, surgical interventions on the chest organs, and pronounced artifacts on CT scans that prevent an adequate lung lesion assessment and measurement of the great vessel diameters.
The non-inclusion criterion is the absence of viral pneumonia signs on CT scans (CT–0) due to a limited number of such admissions.
Setting
The study was conducted with a group of patients who were treated at the temporary reserve hospital in Krylatskoye Ice Palace (Vorokhobov City Clinical Hospital No. 67) from October 08, 2020, to December 05, 2020.
Medical intervention
in a specific period, 2998 non-contrast chest CT scans were performed in the hospital for diagnosis or follow-up of patients with COVID-19 viral pneumonia.
Fig. 2. Airo TruCT mobile CT scanner was installed in the admission department of the temporary reserve hospital for the treatment of patients with COVID-19, which was organized in the Krylatskoe Ice Palace.
CT scans were performed on an Airo TruCT mobile CT scanner (Stryker, USA), which was installed at the reserve hospital for the treatment of patients with COVID-19 (Fig. 2). The mobile CT scanner has an extended aperture diameter of 105 cm and 32 detector rows. This CT system was designed for neurosurgical operating rooms. However, its compact size and low power requirements (possibility of connection to the 1.5 kW power grids) allows an effective usage of the device in a temporary hospital setting. In addition, the power supply unit of the tomograph allows short-term scanning with a power of up to 30 kV. Given the necessity of providing around-the-clock operation mode, the following scanning parameters were chosen: X-ray tube voltage: 120 kV, current: 38 mA, slice thickness:1 mm, matrix: 512×512, pitch: 1.415, rotation time: 1.92 sec, average scanning length: 30 cm (12 sec), and effective dose: 3.9 mSv. Maximum flow is up to 6 patients per hour.
The anonymized CT scans were independently assessed by two radiologists with 3 and 9 years of experience, respectively. The software of the Unified Radiological Information Service of the Unified Medical Information and Analytical System of Moscow City was used on the Agfa Enterprise Imaging platform (Agfa HealthCare, Belgium) to view CT scans and perform measurements. Axial CT 1 mm slices with pulmonary and soft-tissue viewing window parameters, (window width and level of 1500 HU, -500 HU, 350 HU, and 50 HU, respectively) were used for lung lesion degree assessment and great vessel measurement.
With a large patient flow, an empirical visual scale was used to assess the lung lesions based on the visual assessment of the thickened lung tissue volume according to axial and reconstructed chest coronal and sagittal images. Computer-assisted methods of assessing the affected parenchyma were not used.
The lung lesions, according to grades 1–4 of the CT classification, were assessed according to the approved guidelines [1, 2] and the scheme for describing pathological changes, which was proposed by domestic authors, based on the indication of the pulmonary parenchymal lesion volume. Pulmonary tissue changes (frosted glass, reticular thickening, consolidation, “cobblestone” appearance, “air bronchogram,” and inverse “halo”) were determined with the assessment of the lesion volume and localization of changes by the lung lobes and segments.
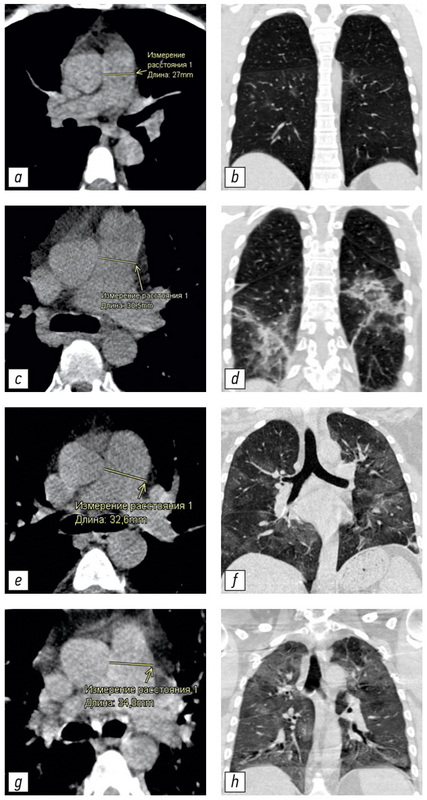
Fig. 3. Examples of CT scans with different PA diameters and lung lesion degrees: a, b, non-dilated PA (27.0 mm) with CT-1 lung lesion (<25%); c, d, dilated PA (30.5 mm) with CT-2 lung lesion (25%–50%); e, f, dilated PA (32.6 mm) with CT-3 lung lesion (50%–75%); g, h, dilated PA (34.8 mm) with CT-4 lung lesion (over 75%).
CT: computed tomography; CT 1–4: adapted scale for visual assessment of the relationship between the patient’s general condition severity and the nature and intensity of radiological signs of pulmonary changes in COVID-19; PA: pulmonary artery.
The PA size was measured on axial sections at the widest point, perpendicular to the long axis of the vessel (Fig. 3, a, c, e, g). The Asc Ao size was measured at the level of the maximum diameter of the right PA [13]. The accuracy of measurements was limited by the absence of contrast enhancement, cardiac gating, and reduced signal-to-noise ratio. Results were obtained by averaging independent measurements taken by two radiologists. A significant difference in the results of parenchymal lesion assessment or great vessel measurement (>2 mm) considers the opinion of a third radiologist with 15 years of work experience.
Main study outcome
The main study outcome is an assessment of the correlation between various measured PA and Asc Ao parameters and their derivative value with pulmonary parenchymal lesions and age dependence.
Additional study outcomes
An additional study outcome is an assessment of the relationship between the Asc Ao diameter and the patient’s age, which confirm the previously established patterns.
Subgroup analyses
In the course of the study, patients of both genders were divided into four groups depending on the lung lesion volume (CT 1–4) and three age ranges (up to 50, 51–69, and over 70 years).
Ethical review
The study was conducted under the Declaration of Helsinki, which was developed by the World Medical Association.
The study was approved by the Independent Ethics Committee of the Moscow Regional Branch of the Russian Society of Roentgenologists and Radiologists, No. 2/2021, dated February 18, 2021.
Informed consent for medical intervention (CT examination) and the use of their medical data for scientific purposes was obtained from all participants.
Statistical analysis
Statistical data processing was conducted using StatSoft Statistica 12.0 software. The required sample size was determined using Altman’s nomogram (with a power of 0.8 and a significance level of 0.05). Examination of over 500 patients obtained the minimum sufficient sample size and statistically significant results. The quantitative data distribution normality was checked using the Kolmogorov–Smirnov criterion and adjusted for Lilliefors and Shapiro–Wilk tests. Given the non-normal distribution of PA and Asc Ao parameters in several studied groups and different numbers of patients divided into groups, the reliability of differences was assessed using the nonparametric Kruskal–Wallis (K-W) test, nonparametric ANOVA analog, and median test (MT). The nonparametric Spearman’s correlation coefficient (ρ) was used to further assess the relationship of several studied parameters of great vessel sizes, whereas 2 × 2 tables and nonparametric Fisher F-criterion were used to analyze differences between the groups. The significance level (p) in all tests was <0.05.
RESULTS
Study subjects/participants
CT scans of 511 patients both male (52.2%) and female (47.8%), with a mean age of 57 ± 12 years, the median age of 59 years, IQR of 49.0–65.0 years, min. of 31 years, and max. of 84 years, were analyzed. The distribution of patients by gender, lung lesion degree, and age intervals is shown in Table 1. Patient CT scans with different pulmonary parenchymal lesions and corresponding PA measurements are presented in Fig. 3.
Table 1. Distribution of patients in groups by gender (M: males), age, and severity of pneumonia according to the CT 1-4 scale
Parameters | CT-1 | CT-2 | CT-3 | CT-4 | Total, n |
Group I (up to 50 years old) | 47 (34 М) | 56 (38 М) | 19 (14 М) | 9 (9 М) | 131 (95 М) |
Group II (50–69 years old) | 84 (36 М) | 127 (56 М) | 78 (39 М) | 24 (19 М) | 313 (150 М) |
Group III (over 70 years old) | 24 (8 М) | 18 (4 М) | 16 (4 М) | 9 (6 М) | 67 (22 М) |
Total, n | 155 (78 М) | 201 (98 М) | 113 (57 М) | 42 (34 М) | 511 (267 М) |
Main study results
The following significant patterns were obtained: an increased PA diameter with lung lesion severity (K-W, p < 0.001; MT, p < 0.001), increasd Asc Ao diameter with the patient’s age (K-W, p < 0.001; MT, p < 0.001), increased PA/Asc Ao ratio with lung lesion severity (K-W, p < 0.001; MT, p = 0.008), and decreased parameter with age (K-W, p < 0.001; MT, p < 0.001). Both the correlation of a small increase in the pulmonary trunk diameter with age (K-W, p = 0.094; MT, p = 0.311) and a small increase in the aortic diameter depending on lung lesion degree (K-W, p = 0.061; MT, p = 0.165) were not significant.
Fig. 4. Dependence of the PA diameter on age and lung lesion severity in COVID-19
CT 1–4: adapted scale for visual assessment of the relationship between the patient’s general condition severity and the nature and intensity of radiological signs of pulmonary changes in COVID-19; PA: pulmonary artery.
Results presented in Fig. 4 determined the following Spearman’s correlation coefficients (between the PA diameter and lesion severity): for the entire sample, ρ = 0.268, p < 0.005; up to 50 years, ρ = 0.245, p < 0.005; 50–70 years, ρ = 0.229, p < 0.005; and over 70 years, ρ = 0.374, p < 0.005, thus a weak positive relationship was noted. The analysis of differences by Fisher’s one-sided F-criterion showed reliable PA dilation (>29 mm) in patients with severe pneumonia (CT 3–4) (Table 2).
Table 2. Distribution of examined patients by age interval, pneumonia severity, and pulmonary artery dilation
Age interval, years | Parameters | |||
CT severity | PA < 29 mm | PA ≥ 29 mm | PA ≥ 29 mm, % | |
Up to 50* | CT 3–4 | 10 | 18 | 64,3 |
CT 1–2 | 80 | 23 | 22,3 | |
50–69** | CT 3–4 | 47 | 51 | 52,0 |
CT 1–2 | 145 | 62 | 29,9 | |
Over 70*** | CT 3–4 | 10 | 19 | 65,5 |
CT 1–2 | 30 | 16 | 34,8 | |
Note. The differences are significant: *p < 0.0001, **p = 0.0001, and ***p = 0.0089 (Fisher’s F-criterion). CT: computed tomography; CT 1–4: adapted scale for visual assessment of the relationship between the patient’s general condition severity and the nature and intensity of radiological signs of pulmonary changes in COVID-19; PA: pulmonary artery.
Additional study results
The following additional study results were obtained: a more frequently (37%) observed severe COVID-19 (CT 3–4) in the older patient group (>70 years); less frequently (21%) observed in the second age group (<50 years); and intermediately (32%) observed in the middle age group (50–69 years old).
In addition, an increased PA diameter from CT–1 to CT–4 was 12.9% (calculated as the ratio of the difference in values to half of the sum), whereas the ratio of the standard deviation to the mean PA values was 13.5%. The relative increase of the PA/Asc Ao ratio was 8.3% and the ratio of the standard deviation to the mean was 11.9%. Therefore, an increased PA diameter was more pronouncedly associated with an increased CT degree compared to the PA/Asc Ao ratio with a smaller difference in the coefficient of variation. In addition, a separate PA changes assessment by this parameter in contrast to the PA/Asc Ao ratio is more preferable.
Adverse events
No adverse events were observed in patients when performing the non-contrast chest CT. Scans were obtained according to the standard non-contrast protocol for chest disease diagnoses.
No adverse events associated with the use of the Airo TruCT mobile CT scanner were registered. The radiation exposure did not exceed the typical values for stationary computed tomographs.
DISCUSSION
Main study result summary
The relationships of PA and Asc Ao diameters and their ratios in patients with different COVID-19-associated pneumonia severity depending on age were studied. A significant positive correlation between the pulmonary trunk diameter and the pulmonary parenchymal lesion in COVID-19 was revealed. A non-significant increased pulmonary trunk diameter with age was shown. A significant aortic dilatation was observed in older age groups, without relation to the increasing disease severity.
Main study result discussion
An equal but not significant increased pulmonary trunk diameter with age (see Fig. 4) was shown. Values for age intervals up to 50, 50–69, and over 70 years amounted for 27.0 ± 3.6, 27.6 ± 3.8, and 28.2 ± 4.1 mm (mean ± standard deviation), respectively. However, a significantly increased PA diameter was shown about pulmonary parenchymal lesion severity in COVID-19-associated viral pneumonia (see Fig. 4). CT grades 1–4 determined the following mean PA values: 26.7 ± 3.4, 26.9 ± 3.4, 28.8 ± 4.1, and 30.4 ± 4.4 mm, respectively. Reverse patterns were shown for the aortic diameter. In addition, a significant change (increase) in the aortic diameter with age (31.2 ± 3.7, 34.6 ± 3.9, and 34.8 ± 5.6 mm) for age intervals defined above was observed. No significant change (increase) was noted in the aortic diameter with increasing lung lesion severity (CT 1–4), that is, 33.3 ± 4.9, 33.7 ± 4.2, 34.2 ± 3.9, and 34.6 ± 4.3 mm, respectively. The derived parameter (PA/Asc Ao ratio) showed a significant change (decrease) with age (0.87 ± 0.11, 0.80 ± 0.12, and 0.82 ± 0.15 RU, respectively) due to a pronounced increased Asc Ao diameter with age and a slight increased PA diameter. A significant change (increase) in the PA/Asc Ao ratio with increasing degree of the pulmonary tissue lesion (CT 1–4) was shown, that is 0.81 ± 0.13, 0.81 ± 0.11, 0.85 ± 0.14, and 0.88 ± 0.12 RU, respectively.
The obtained mean PA diameter and PA/Asc Ao ratio in the CT–1 group with the smallest lesion volume (26.7 ± 3.4 and 0.81 ± 0.13) were slightly higher compared with the mean values defined as normal in the Framingham Heart Study [14], where the mean PA size was 25.1 ± 2.8 mm and the mean PA/Asc Ao ratio was 0.77 ± 0.09. This is due to the methodological peculiarities, as measurements were performed without cardiac gating, which resulted in increased measured vessel diameters and disproportionate division of the studied groups by gender. The mean measured aortic diameter in the group of patients up to 50 years old (31.2 ± 3.7 mm) was well comparable with the review results [15], which is devoted to age-related aortic measurements (31.1 ± 4 mm). In addition, this review noted an increased aortic diameter with age due to decreased vessel wall elasticity.
The groups with severe COVID-2019 (CT 3–4) (see Fig. 2, b, d, f, h) demonstrated a statistically significant increased proportion of patients with PA diameter dilation of >29 mm for all ages (Table 2). On average, this group of patients was 60.6%, which was twice the average number of patients with PA dilation of >29 mm in the groups with mild COVID-19 (CT 1–2).
Our data agree well with the previously obtained results [9], where PA dilation in patients with COVID-19-associated pneumonia compared to the data before the disease showed a significantly increased PA/Asc Ao ratio. However, the relationship of these parameters with the pneumonia severity was not pronounced.
The threshold PA diameter values, above which the pulmonary hypertension is reliably stated, were different. For example, the upper limits of the PA diameter, above which patients had dyspnea, were 29 mm in males and 27 mm in females, whereas the critical PA/Asc Ao ratio was 0.9 [13, 16]. Another study proposed PA values of 29 mm and PA/Asc Ao ratio as criteria above which pulmonary hypertension is diagnosed. However, these values showed high sensitivity and insufficient specificity [14]. Alternatively, another proposed threshold of 31.5 mm, above which pulmonary hypertension is registered, showed low specificity [14]. The guidelines for pulmonary hypertension diagnosis and treatment noted a threshold of 29 mm [17].
Thus, the chosen threshold of 29 mm is reasonable. However, this value will probably be adjusted for the additional PA pressure assessment data, which is obtained by ultrasound measurements [18]. The statistical data on a significantly higher number of patients with a PA diameter of >29 mm in groups with a severe course (see Table 2) indicate a possible contribution of pulmonary hypertension to a more severe disease course.
The PA/Asc Ao ratio did not demonstrate threshold values exceeding 0.9 and 1 [9] and reached maximum values of 0.88 ± 0.12 in the group with a CT-4 degree of lung lesions. A significant increase of this index corresponds to the results reported by P. Spagnolo et al. [9]. While emphasizing the clinical significance of PA dilation, the authors provided reliable data on increased PA diameter and PA diameter/Asc Ao ratio in patients with lethal outcomes compared to those who recovered. This study revealed a weak positive correlation between the lung lesion degree (also divided into severity grades 1–4) and an increased PA diameter. A significant PA diameter dilation was shown in the examined patients compared to the state before the development of pneumonia; however, the sample size was small (45 patients). The necessity of PA dilation correction with PA/Asc Ao ratio determination is confirmed by a reliably smaller, but rather high number of patients with PA dilation in the cohort with small lesion volume (up to 50%) (Table 2). The use of this criterion is reasonable for constitutionally excluding conditioned wide PA. However, this auxiliary comparative criterion is inefficient in patients over 50 years old with hypertension since the aorta also expands due to the vessel wall connective tissue damage [15].
The effectiveness of the PA measurement as a prognostic sign of pulmonary hypertension and an additional severity marker is confirmed by the preferential bilateral process of pulmonary tissue lesions [9]. The PA accumulates increasing hemodynamic distress in the small circle as an additional predictor of COVID-19-associated viral pneumonia severity. PA dilation, which was a reliable relationship with parenchymal lesions, resulted from increasing pulmonary hypertension.
Pulmonary autoimmune diseases show signs of interstitial pneumonia, and pulmonary hypertension develops, for example, with progressing idiopathic pulmonary fibrosis due to massive interstitial lung lesions [19]. The normal architectonics of the pulmonary vessels is deformed due to the massive pulmonary interstitium inflammation. Capillary constriction and endothelium integrity failure lead to small vessel microthrombosis and sclerosis and, consequently, to an increased hydraulic resistance, which results in pulmonary hypertension [20]. The development of COVID-19-associated viral pneumonia is due to high levels of expression of angiotensin-converting enzyme type 2 (ACE2) in type II alveolar pulmonary cells, which are targets of the spike antigen of severe acute respiratory syndrome coronavirus 2 virus that leads to interstitial lung lesions [11, 12]. The affecting factor that leads to microthrombosis is both a direct vascular endothelium lesion due to ACE2 receptor expression [15, 20, 21] and a possible secondary vascular bed immune lesion and the development of edema and exudation. This is facilitated by the extensive accumulation of complement components in the alveolar walls, which results in endothelial cell damage to the pulmonary capillaries and subsequent coagulation activation [20]. ACE2 receptor system depletion in the pulmonary vascular bed contributes to endothelial dysfunction and inflammation and aggravates the course of atherosclerosis and diabetic angiopathy [15, 20]. In COVID-19, this process leads to delayed adverse reactions in the form of progressing atherosclerotic vascular lesions.
Moreover, the phenomenon of vascular enlargement sign that is a sign of dilated vessels in COVID-19 pneumonia was reported. However, this phenomenon was not deeply studied, and several available studies showed its inconsistent prevalence. This sign occurs in 76.9% of patients with viral pneumonia [7, 10].
Pulmonary hypertension, regardless of its causes, has a secondary negative effect on the course of viral pneumonia, especially with massive lung lesions.
The pulmonary trunk dilation, depending on the severity of pulmonary parenchymal lesions, allows pulmonary hypertension consideration as one of the important signs of COVID-19-associated pneumonia severity. The established pattern allows a more differentiated approach in predicting outcomes of this infectious disease, developing effective therapy methods, and identifying population risk groups for a severe disease course that require increased attention from clinicians.
Study limitations
The study does not answer the question of whether pulmonary hypertension, which is noted as PA dilation, is an initial condition or whether it develops within the course of coronavirus infection. This study did not measure the PA diameter in patients of different ages without signs of viral pneumonia to assess the population prevalence of pulmonary hypertension (PA dilation). Data on the population prevalence of pulmonary hypertension and PA dilation are presented in the mentioned study [20].
CONCLUSIONS
The study showed a statistically significant positive relationship between PA dilation and lung lesion severity in COVID-19-associated pneumonia in all age groups. A higher number of patients with PA dilation of ≥29 mm (signs of pulmonary hypertension) and a severe degree of pulmonary parenchymal lesions (CT 3–4) is statistically significant. The obtained patterns require further study, which allows a more differentiated approach in predicting outcomes of this infectious disease, developing effective therapy methods, and identifying population risk groups for a severe disease course that require increased clinician attention.
ADDITIONAL INFORMATION
Funding source. This study was not supported by any external sources of funding.
Competing interests. The authors declare that they have no competing interests.
Authors’ contribution. A.F. Aliyev ― research idea and design, data collection, processing and analysis; N.D. Kudryavtsev ― research design, data collection, processing and analysis; A.V. Petryaykin ― research design, data processing and analysis; Z.R. Artyukova ― data processing; A.S. Skoda, S.P. Morozov ― research design, data analysis. All authors made a substantial contribution to the conception of the work, acquisition, analysis, interpretation of data for the work, drafting and revising the work, final approval of the version to be published and agree to be accountable for all aspects of the work.
Acknowledgments. We express our gratitude to Fuzuli K. Aliyev, Doctor of Physical and Mathematical Sciences and Tatiana I. Aliyeva, Candidate of Pedagogical Sciences for help in statistical data processing, primary focus of patients’ groups and advice on study design.
About the authors
Alexander F. Aliev
L.A. Vorokhobov Municipal Clinical Hospital No 67 of the Moscow Health Department; The Moscow Research and Clinical Center for Tuberculosis Control of the Moscow Health Department
Email: alijealex83@gmail.com
ORCID iD: 0000-0003-3282-0567
SPIN-code: 7891-9314
MD, Cand. Sci. (Med.)
Russian Federation, 2/44, Salyama Adilya street, 123423 Moscow; 10 c1, Stromynka Street, 107014 MoscowNikita D. Kudryavtsev
L.A. Vorokhobov Municipal Clinical Hospital No 67 of the Moscow Health Department; Moscow Center for Diagnostics and Telemedicine
Email: n.kudryavtsev@npcmr.ru
ORCID iD: 0000-0003-4203-0630
SPIN-code: 1125-8637
diagnostic radiologist, junior researcher
Russian Federation, 2/44, Salyama Adilya street, 123423 Moscow; 24 с1, Petrovka street, 127051 MoscowAlexey V. Petraikin
Moscow Center for Diagnostics and Telemedicine
Email: alexeypetraikin@gmail.com
ORCID iD: 0000-0003-1694-4682
SPIN-code: 6193-1656
MD, Cand. Sci. (Med.), Associate Professor, leading researcher, diagnostic radiologist
Russian Federation, 24 с1, Petrovka street, 127051 MoscowZlata R. Artyukova
Moscow Center for Diagnostics and Telemedicine
Email: z.artyukova@npcmr.ru
ORCID iD: 0000-0003-2960-9787
SPIN-code: 7550-2441
MD, junior researcher, radiologist
Russian Federation, 24 с1, Petrovka street, 127051 MoscowAndrey S. Shkoda
Vorokhobov City Clinical Hospital No. 67
Email: gkb67@zdrav.mos.ru
ORCID iD: 0000-0002-9783-1796
MD, Dr. Sci. (Med.), Professor, doctor of the highest qualification category in the specialty, chief physician
Russian Federation, 2/44, Salama Adil street, 123423 MoscowSergey P. Morozov
Moscow Center for Diagnostics and Telemedicine
Author for correspondence.
Email: morozov@npcmr.ru
ORCID iD: 0000-0001-6545-6170
SPIN-code: 8542-1720
MD, Dr. Sci. (Med.), Professor
Russian Federation, 24 с1, Petrovka street, 127051 MoscowReferences
- Morozov SP, Protsenko DN, Smetanina SV, et al. Radiation diagnostics of coronavirus disease (COVID-19): organization, methodology, interpretation of results: preprint No. CDT-2020-II. Version 2 from 17.04.2020. The series "Best practices of radiation and instrumental diagnostics". Issue 65. Moscow: Scientific and Practical Clinical Center for Diagnostics and Telemedicine Technologies of the Department of Healthcare of the City of Moscow; 2020. 78 p. (In Russ).
- Prevention, diagnosis and treatment of a new coronavirus infection (COVID-19): Temporary methodological recommendations. Version 10 (08.02.2020). 2020. 261 p. (In Russ).
- Fomin VV, Ternovoy SK, Serova NS. Radiological guidelines in patients with COVID-19 (Sechenov University experience). REJR. 2020;10(2):8–13. (In Russ). doi: 21569/2222-7415-2020-10-2-8-13
- Henkel M, Weikert T, Marston K, et al. Lethal COVID-19: radiological-pathological correlation of the lungs. Radiol Cardiothorac Imaging. 2020;2(6):e200406. doi: 10.1148/ryct.2020200406
- Sun Z, Zhang N, Li Y, et al. A systematic review of chest imaging findings in COVID-19. Quant Imaging Med Surg. 2020;10(5): 1058–1079. doi: 10.21037/qims-20-564
- Salehi S, Abedi A, Balakrishnan S, et al. Coronavirus disease 2019 (COVID-19): A systematic review of imaging findings in 919 patients. AJR Am J Roentgenol. 2020;215(1):87–93. doi: 10.2214/AJR.20.23034
- Qanadli SD, Beigelman-Aubry C, Rotzinger DC. Vascular changes detected with thoracic CT in coronavirus disease (COVID-19) might be significant determinants for accurate diagnosis and optimal patient management. AJR Am J Roentgenol. 2020;215(1):15. doi: 10.2214/AJR.20.23185
- Li X, Ma X. Acute respiratory failure in COVID-19: Is it "typical" ARDS? Crit Care. 2020;24(1):198. doi: 10.1186/s13054-020-02911-9
- Spagnolo P, Cozzi A, Foà RA, et al. CT-derived pulmonary vascular metrics and clinical outcome in COVID-19 patients. Quant Imaging Med Surg. 2020;10(6):1325–1333. doi: 10.21037/qims-20-546
- Lv H, Chen T, Pan Y, et al. Pulmonary vascular enlargement on thoracic CT for diagnosis and differential diagnosis of COVID-19: a systematic review and meta-analysis. Ann Transl Med. 2020; 8(14):878–878. doi: 10.21037/atm-20-4955
- Chang YC, Yu CJ, Chang SC, et al. Pulmonary sequelae in convalescent patients after severe acute respiratory syndrome: Evaluation with thin-section CT. Radiology. 2005;236(3):1067–1075. doi: 10.1148/radiol.2363040958
- Prokop M, van Everdingen W, van Rees Vellinga T, et al. CO-RADS: A categorical CT assessment scheme for patients suspected of having COVID-19-definition and evaluation. Radiology. 2020;296(2):E97–E104. doi: 10.1148/radiol.2020201473
- Corson N, Armato SG, Labby ZE, et al. CT-based pulmonary artery measurements for the assessment of pulmonary hypertension. Acad Radiol. 2014;21(4):523–530. doi: 10.1016/j.acra.2013.12.015
- Truong QA, Massaro JM, Rogers IS, et al. Reference values for normal pulmonary artery dimensions by noncontrast cardiac computed tomography the framingham heart study. Circ Cardiovasc Imaging. 2012;5(1):147–154. doi: 10.1161/CIRCIMAGING.111.968610
- Collins JA, Munoz JV, Patel TR, et al. The anatomy of the aging aorta. Clin Anat. 2014;27(3):463–466. doi: 10.1002/ca.22384
- Compton GL, Florence J, MacDonald C, et al. Main pulmonary artery-to-ascending aorta diameter ratio in healthy children on MDCT. AJR Am J Roentgenol. 2015;205(6):1322–1325. doi: 10.2214/AJR.15.14301
- Galiè N, Humbert M, Vachiery JL, et al. 2015 ESC/ERS guidelines for the diagnosis and treatment of pulmonary hypertension: the joint task force for the diagnosis and treatment of pulmonary hypertension of the European society of cardiology (ESC) and the European Respiratory Society (ERS): Endorsed by: Association for European Paediatric and Congenital Cardiology (AEPC), International Society for Heart and Lung Transplantation (ISHLT). European Heart Journal. 2016;37(1):67–119. doi: 10.1093/eurheartj/ehv317
- Parasuraman S, Walker S, Loudon BL, et al. Assessment of pulmonary artery pressure by echocardiography — A comprehensive review. Int J Cardiol Heart Vasc. 2016;12:45–51. doi: 10.1016/j.ijcha.2016.05.011
- Chuchalin AG, Avdeev SN, Aysanov ZR, et al. Diagnosis and treatment of idiopathic pulmonary fibrosis federal clinical guidelines. Pulmonology. 2016;26(4):399–419. (In Russ). doi: 10.18093/0869-0189-2016-26-4-399-419
- Chernyaev AL, Samsonova MV. Pathological anatomy of the lungs. Atlas. 2nd ed., revised and updated. A series of monographs of the Russian Respiratory Society. Ed. by A.G. Chuchalin. Moscow: Atmosfera; 2011. 111 p. (In Russ).
- Dolhnikoff M, Duarte-Neto AN, de Almeida Monteiro RA, et al. Pathological evidence of pulmonary thrombotic phenomena in severe COVID-19. J Thromb Haemost. 2020;18(6):1517–1519. doi: 10.1111/jth.14844
Supplementary files