Polyostotic fibrous dysplasia: imaging findings of a controversial case
- Authors: De Michele F.1,2, Carpagnano F.A.1,2, Paparella M.T.1,2, Guglielmi G.1,2,3
-
Affiliations:
- Department of Clinical and Experimental Medicine, Foggia University School of Medicine
- Radiology Unit, Barletta University Campus UNIFG, “Dimiccoli” Hospital
- Radiology Unit, Hospital “Casa Sollievo Della Sofferenza”, San Giovanni Rotondo
- Issue: Vol 3, No 1 (2022)
- Pages: 55-63
- Section: Case reports
- Submitted: 17.11.2021
- Accepted: 17.01.2022
- Published: 24.04.2022
- URL: https://jdigitaldiagnostics.com/DD/article/view/88605
- DOI: https://doi.org/10.17816/DD88605
- ID: 88605
Cite item
Abstract
Fibrous dysplasia is a rare non-neoplastic tumor-like congenital bone disease that is most likely associated with GNAS gene mutations, with a broad spectrum of clinical presentations, ranging from isolated monostotic and polyostotic forms to other extra-skeletal associated manifestations as in McCune–Albright syndrome. It is responsible for bone’s weakening and increased fragility, making it prone to fractures.
A 65-year-old female patient was referred to our radiology department for cervical and dorsal pain, with a previous diagnosis of incidental cervical and dorsal bone lesions that are suspected for metastases. X-ray, computed tomography, and magnetic resonance imaging were performed with a precise diagnostic suspicion of fibrous dysplasia that is confirmed by bone biopsy.
Fibrous dysplasia principally affects the bone and is characterized by bone replacement itself by dysplastic fibrous tissue. According to the number of affected bones and their association to endocrine alterations, it is classified into three categories monostotic, polyostotic, and Albright’s disease. Differential diagnosis with multiple myeloma among others and the best treatment decision was made.
Full Text
BACKGROUND
Fibrous dysplasia (FD) is a benign progressive condition that is characterized by abnormal fibroblast proliferation and bone and marrow replacement with fibrous connective tissue [1]. Described for the first time by von Recklinghausen in 1891 [2], FD is a rare congenital disease caused by a sporadic postzygotic missense mutation that leads to abnormal fibroblast proliferation, defective osteoblast differentiation, and increased bone resorption [3-4]. It may affect one or several bones, according to the monostotic or polyostotic type, and may be associated with hyperfunction endocrinopathies and hyperpigmentation of the skin (café-au-lait spots) in the so-called “McCune–Albright syndrome” (MAS) [5-6]. FD has a predilection for certain bones including the femur, tibia, ribs, pelvis, and craniofacial. The estimated prevalence of FD is 1/30,000, with the monostotic type 5–6 times as prevalent as the polyostotic one. Generally, FD is a silent disease and some patients are asymptomatic. With the presence of symptoms, they often include bone pain, headache, bone deformities, facial asymmetry, proptosis, diplopia, pathological fractures, and during childhood. More serious symptoms include optic neuropathy, blindness, deafness, vestibular dysfunction, and occasionally, facial nerve palsy. Scintigraphy/isotopic bone scans are normally used to assess the extent of FD and the number of FD lesions, whereas computed tomography (CT) and, even less, magnetic resonance imaging (MRI) are useful to characterize the typical morpho-structural characteristics of each bone lesion, which usually have a lytic appearance and are characterized by thinning of the cortex with a “ground-glass” matrix [7]. Biochemical markers of bone turnover are in some cases elevated and final diagnosis can be confirmed by biopsy. Moreover, hypophosphatemia may be present together with increased secretion of fibroblast growth factor (FGF-23) from the dysplastic tissue [8]. Hypophosphatemia may contribute to mineralization defects, which can lead to osteomalacia, rickets, and bone pain with deformities [9]. Cure or spontaneous resolution of FD are unavailable, but not all patients require treatment. Surgery may be indicated for larger lesions, which cause adjacent tissue compression with nerve constriction and pain. Bisphosphonates are often used as a medical treatment as they have been suggested to reduce the increased bone resorption at the affected sites.
DESCRIPTION OF THE CASE
Clinical data
Herein, presented a case of a 65-year-old female patient, without a specific clinical history, who was referred to our radiology department for cervical pain and discomfort for some years, as the only symptom. There was no history of preceding trauma, but the physical examination revealed dark-colored spots on the skin (café-au-lait spots), facial asymmetry, and obesity. In the previous years, she underwent several diagnostic exams to exclude attributable pathologies to her symptoms, resulting in the presence of lytic lesions of the cervical and dorsal metamers. Therefore, she was investigated for the suspicion of bone heteroplasia or neoplastic secondarysm, without an exhaustive final diagnosis. When she was referred to our radiology department, we decided to investigate her multiple known bone lesions from a multi-diagnostic perspective. Therefore, we performed an X-ray, CT, and MRI scan of the skeleton, with particular attention on segments that are affected by the above-mentioned signs and symptoms. Then we required positron emission tomography (PET) - and Single-photon emission computed tomography (SPECT) scans, as well as a bone biopsy, to confirm our diagnostic suspicion.
Radiological findings
X-ray imaging showed the presence of well-circumscribed lytic lesions at the level of C5, C7, and D1 vertebrae and at the posterior arch of the fifth left rib, together with a larger localization of the disease at the first left rib’s posterior arch, which presented a bubbly cystic bone lesion with a cortical “blown appearance”, cortical thinning, and none periosteal reaction (Fig. 1).
Fig. 1. The cervical tract X-ray, in the context of a diffused uncovertebral arthrosis, shows the presence of some ill-defined lithic bony lesions at C5, C7, and D1 vertebrae (a) and the posterior arch of the fifth left rib (b); moreover, a 5.5 × 2.5 cm expansile lesion of the posterior first left rib is visible, with a swollen and blown cortical appearance (b).
The multiplanar reconstruction CT scan showed a “ground-glass” appearance of the bone matrix, together with homogeneously sclerotic and lytic areas of involved cervical vertebrae, underlying the involvement of C7 and D1 metamers and confirming the C5’s one (Fig. 2a, 2b, and 2c).
The first posterior left rib, already described with a plain radiograph, was better investigated, resulting in the presence of a huge expansile bone lesion with a thin overlying cortical bone and “ground-glass” bone matrix appearance together with cystic lytic areas, which confirm the complete substitution of the trabecular bone with fibrous tissue (Fig. 2d).
Moreover, the total-body CT scan showed the involvement of the fifth dorsal vertebra along the posterior corresponding left rib, which shows the same bony changes as others (Fig. 2e).
Fig. 2. Sagittal (a) and coronal (b) cervical CT showing extensive lytic bone lesions at C5, C7, and D1 vertebrae, involving the seventh cervical posterior laminae and spinous process (c). The posterior arch of the first left rib shows an expansile bone lesion, surrounded by a thin and blown cortical layer, with a ground-glass bone matrix appearance and no soft tissue component (better seen on the axial plane in the figure 2d). Axial plane of the thoracic CT scan showing the involvement of the fifth dorsal vertebra, together with the corresponding left rib (e).
The MRI, even if not straight and not particularly useful for the differential diagnosis, was performed, thus confirming the polyostotic skeletal involvement.
It showed T1 hypointense lytic lesions at the level of C5, C7, D1, D4, and D5 vertebrae, with involvement of posterior elements of C5–C6 (Fig. 3a) and a corresponding heterogeneous contrast enhancement in T1w sequences after Gadolinium administration (Fig. 3b). The bone tissue showed extensive remodeled areas, without confirmed periosteal reaction.
Fig. 3. Sagittal T1w sequence of the cervical tract shows heterogeneously low-signal bone lytic lesions involving C5, C7, D1, D4, and D5 vertebrae and the spinous processes of C5 and C6 metamers (3a); all these findings were also confirmed by the administration of Gadolinium contrast enhancement (3b).
Nuclear medicine
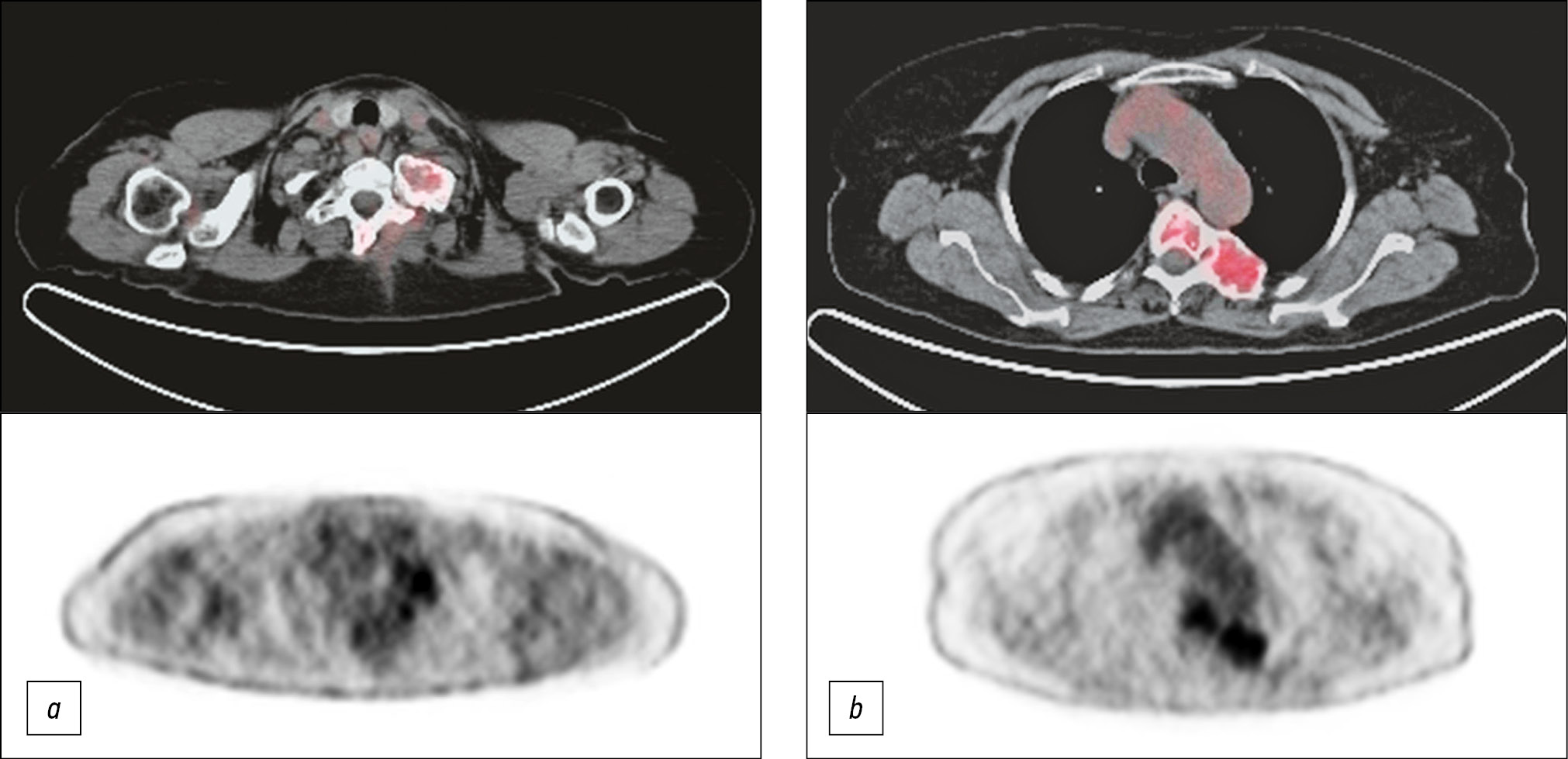
The whole body Fluorine-18 Fluorodeoxyglucose PET-CT scan confirmed the involvement of each of the above-mentioned skeletal segments and identified with other imaging methods but, paying special attention to some of them that showed more intense uptake of the radiotracer, specifically the posterior arch of the first left rib, the fifth vertebral body, and the left posterior arch of the corresponding rib (Fig. 4).
Fig. 4. 18F-FDG PET-CT scan showing an increased tracer uptake at the posterior arch of the first left rib (4a), the fifth dorsal vertebra, and the posterior arch of the corresponding left rib (4b).
Additionally, the presence of a marked metabolic activity at these levels was confirmed by the SPECT-CT scan, which showed two areas of intense and pathological focal accumulation of the osteotropic tracer (99mTc-HDP) in the left paravertebral side. The upper one, corresponds to the first dorsal vertebra, with an SUV max of 39.91, and the other at D5-fifth left rib with an SUV max of 47.06 (Fig. 5).
Fig. 5. The whole body SPECT-CT confirmed the involvement of the above-mentioned bone segments and no other particularly shows two areas of high pathological focal accumulation of the osteotropic tracer (99mTc-HDP) in the left paravertebral side (D1 and D5-fifth left rib).
Biopsy
Bone biopsy was performed, which revealed the presence of osteoporotic and necrotic bone spicules with cartilagineous and fibromuscular fragments in extensive myxoid areas. Thus, the diagnosis of polyostotic FD was made, which was a very important step in the diagnostic process because the neoplastic nature of the lesions was excluded, unlike assumed by the reports of previous years.
CONCLUSION
FD primary affects the bones, causing the replacement of the osseous matrix by dysplastic fibrous tissue, with a sporadic extra-skeletal involvement. It is classified based on the number of affected bones and its association to endocrine alterations in monostotic, polyostotic, and Albright’s disease. A congenital etiology is suggested and pathologic fractures are the most frequently associated complications.
Herein, presented a case of an adult female patient with a long story of unexplained cervical and dorsal pain.
Thanks to a multidisciplinary approach and integrated imaging, which allowed us to exclude the neoplastic nature of bone injuries, orienting our intuition toward the diagnostic suspicion of polyostotic FD was possible, which was confirmed by histological examination.
ADDITIONAL INFORMATION
Funding source. This article was not supported by any external sources of funding.
Competing interests. The authors declare no explicit and potential conflicts of interest associated with the publication of this article.
Authors’ contribution. All authors made a substantial contribution to the conception of the work, acquisition, analysis, interpretation of data for the work, drafting and revising the work, final approval of the version to be published and agree to be accountable for all aspects of the work.
Consent for publication. Informed written consent was taken from the subject of discussion before submission.
About the authors
Francesca De Michele
Department of Clinical and Experimental Medicine, Foggia University School of Medicine; Radiology Unit, Barletta University Campus UNIFG, “Dimiccoli” Hospital
Email: francesca.demichele82@gmail.com
ORCID iD: 0000-0002-6043-6362
Italy, Foggia; Foggia
Francesca A. Carpagnano
Department of Clinical and Experimental Medicine, Foggia University School of Medicine; Radiology Unit, Barletta University Campus UNIFG, “Dimiccoli” Hospital
Email: c.francesca1991@gmail.com
ORCID iD: 0000-0001-7681-2898
Italy, Foggia; Foggia
Maria T. Paparella
Department of Clinical and Experimental Medicine, Foggia University School of Medicine; Radiology Unit, Barletta University Campus UNIFG, “Dimiccoli” Hospital
Email: mt.paparella@gmail.com
ORCID iD: 0000-0003-2573-9509
Italy, Foggia; Foggia
Giuseppe Guglielmi
Department of Clinical and Experimental Medicine, Foggia University School of Medicine; Radiology Unit, Barletta University Campus UNIFG, “Dimiccoli” Hospital; Radiology Unit, Hospital “Casa Sollievo Della Sofferenza”, San Giovanni Rotondo
Author for correspondence.
Email: giuseppe.guglielmi@unifg.it
ORCID iD: 0000-0002-4325-8330
Medical Doctor, Full Professor of Radiology, Department of Clinical and Experimental Medicine.
Italy, Foggia; Foggia; FoggiaReferences
- Neville BW, Damm DD, Allen CM, Bouquot JE. Bone pathology. In: Neville BW, Damm DD, Allen CM, Bouquot JE, editors. Oral and maxillofacial pathology. Philadelphia, PA: WB Saunders Company; 2002. Р. 553–557.
- Iseri PK, Efendi H, Demirci A, Komsuoglu S. Fibrous dysplasia of the cranial bones: a case report and review of the literature. Yale J Biol Med. 2005;78(3):141–145.
- Lustig LR, Holliday MJ, McCarthy FF, Nager GT. Fibrous dysplasia involving the skull base and temporal bone. Arch Otolaryngol Head Neck Surg. 2001;127(10):1239–1247. doi: 10.1001/archotol.127.10.1239
- Chapurlat RD, Orcel P. Fibrous dysplasia of bone and McCune-Albright syndrome. Best Pract Res Clin Rheumatol. 2008;22(1):55–69. doi: 10.1016/j.berh.2007.11.004
- Feller L, Wood N, Khammissa R, et al. The nature of fibrous dysplasia. Head Face Med. 2009;5:22. doi: 10.1186/1746-160X-5-22
- Riminucci M, Kuznetsov SA, Cherman N, et al. Osteoclastogenesis in fibrous dysplasia of bone: in situ and in vitro analysis of IL-6 expression. Bone. 2003;33(3):434–442. doi: 10.1016/s8756-3282(03)00064-4
- Kransdorf MJ, Moser RP, Gilkey FW. Fibrous dysplasia. Radiographics. 1990;10(3):519–537. doi: 10.1148/radiographics.10.3.2188311
- Bhattacharyya N, Wiench M, Dumitrescu C, et al. Mechanism of FGF23 processing in fibrous dysplasia. J Bone Miner Res. 2012;27(5):1132–1141. doi: 10.1002/jbmr.1546
- Yamamoto T, Imanishi Y, Kinoshita E, et al. The role of fibroblast growth factor 23 for hypophosphatemia and abnormal regulation of vitamin D metabolism in patients with McCune-Albright syndrome. J Bone MinerMetab. 2005;23(3):231–237. doi: 10.1007/s00774-004-0589-9
Supplementary files