Interdisciplinary databank in oncoendocrinology: radioiodine refractory differentiated thyroid cancer
- Authors: Rumyantsev P.O.1, Baidak A.G.2
-
Affiliations:
- International Medical Center “SOGAZ-MEDICINE”
- M-Data
- Issue: Vol 3, No 1 (2022)
- Pages: 86-93
- Section: Editorials
- Submitted: 11.03.2022
- Accepted: 08.04.2022
- Published: 24.04.2022
- URL: https://jdigitaldiagnostics.com/DD/article/view/104745
- DOI: https://doi.org/10.17816/DD104745
- ID: 104745
Cite item
Abstract
High achievements in personalized evidence-based medicine and oncology, particularly in developed countries, are associated with the successful development of “clinical” carceral registries of patients (SEER, NCDB, etc.), which are multimodal data banks. They are a data matrix for the development of analytical and prognostic tools in the study of diagnostic features, clinical disease course, therapy response, assessment of prognostic factors, etc. From the point of view of medical digital data banks, data redundancy and duplication are not as critical as information incompleteness or inconsistency when making medical decisions.
This study aimed to present a multimodal database of patients with radioiodine refractory differentiated thyroid cancer, which is essentially a modern interdisciplinary digital medical registry.
Along with demographic and nosological data that is typical for epidemiological registers, the multimodal data banks consider key clinical and paraclinical data, such as the results of laboratory, morphological, and instrumental research methods, and various imaging methods, such as ultrasound, computed tomography (CT), magnetic resonance imaging, single-photon emission computed tomography/CT, and positron emission tomography/CT. The multimodal data banks present the molecular genetic profile results of the tumor, of which the clinical usefulness in the choice of treatment tactics is undoubted today. All these data are accumulated in the multimodal data banks, noting the execution time and the revision results (second opinion), considering standardized qualitative and quantitative parameters (factors) that potentially affect the clinical course, treatment response, complication development, and outcomes.
Keywords
Full Text
INTRODUCTION
In Russia, the incidence of thyroid cancer (TC) has increased more than twice the number, from 4.41 per 100,000 populations in 1998 to 9.53 in 2019 [1, 2], primarily due to the additional detection of papillary microcarcinomas resulting from widely used ultrasound (US) examinations.
Approximately, 70%–80% of the presently detected thyroid carcinomas have a favorable prognosis, particularly when detected at early stages. Moreover, postoperative iodine-131 therapy is not indicated for this patient group [3]. On average, 10% of patients with differentiated thyroid cancer (DTC) show radioiodine refractoriness (RR) [4].
INTERDISCIPLINARY DATABANKS
At present, registries for patients with cancer (Surveillance, Epidemiology, and End Results, National Cancer Database, and others) or interdisciplinary databanks (IDB) are the major tools worldwide that are used for studying the features of clinical disease course and therapy response and assessing the influence of prognostic factors [5]. The integration of specialists of different profiles on a single information platform allows rapid communication and expert support for medical decision-making [6]. The primary stakeholder for accurate and rapid medical decision-making is the patient; therefore, patient involvement as a “donor” of anamnestic and operative medical information (extracts, tests, disks, and glasses), both in paper and electronic digital formats, is necessary. Redundancy and data duplication are not critical in terms of medical interactive databanks, whereas incomplete or inconsistent information is critical in medical decision-making.
Therefore, the European Union Committee of Experts on Rare Diseases operates to obtain consolidated data on patients who suffer from rare diseases, including rare cancers with an incidence of <6 per 100,000 population and TC also belongs to it [7]. Currently, Russia has several active registries at different healthcare levels.
INTERDISCIPLINARY PATIENT DATABANK PROJECT FOR RADIOIODINE-REFRACTORY DTC
This study aimed to popularize the Interdisciplinary Patient Databank Project for Radioiodine-Refractory Differentiated TC (IDB RR-DTC), which is essentially a modern clinical registry and is supported by the Association of Endocrine Surgeons (https://as-endo.ru/) and the Association for the Development of Theranostics (https://www.theranostics.pro/).
Significant clinical and paraclinical data are considered in the developed IDB RR-DTC, namely, results of laboratory, morphological, and instrumental methods of examination and various imaging techniques (US, computed tomography [CT], magnetic resonance imaging [MRI], single-photon emission CT/CT [SPECT/CT], and positron emission tomography/CT [PET/CT]), in addition to demographic and nosological data characteristic of epidemiological registries. IDB RR-DTC plans to record (when completed) the results of the molecular genetic profile of tumors, which clinical benefit in the choice of treatment tactics is no longer in doubt. All these data will be accumulated in the IDB RR-DTC with a note on the time of execution and revision results (second opinion), including standardized qualitative and quantitative parameters (factors) that potentially influence the clinical course, treatment response, complications, and outcomes.
Several studies showed that tumor growth and TC progression are closely related to somatic point mutations in the BRAF, RAS, and RET genes. These mutations contribute to activating the mitogen-activated protein kinase and phosphoinositide 3-kinase proliferation signaling pathways, which are crucial in TC development [8]. Clinicians should keep in mind that patients with a Bethesda IV, V, and VI cytology report may be prescribed molecular genetic testing in the BRAF, TERT, RAS, RET/PTC, and PAX8/PPAR-γ genes, as recommended by the Ministry of Health of the Russian Federation [9].
The IDB RR-DTC project collects information on molecular genetic testing results for known mutations in the BRAF, TERT, RAS, RET/PTC, and PAX8/PPAR-γ genes for subsequent analysis.
The created IDB RR-DTC uses templates for entering anamnestic data (to show data in one form), editing the existing history, adding dated changes, and attaching documents in PDF format.
Table. Structure of data stored and processed in the Interdisciplinary Patient Databank Project for Radioiodine-Refractory Differentiated Thyroid Cancer (IDB RR-DTC)
Section | Parameters |
Section | Parameters |
Demographics | · Surname, first name, patronymic, date of birth, gender, and address |
History | · Family history · Radiation history (neck irradiation and being in a radiation exposure area) |
Diagnosis(s) | · ICD-10 code · The histological type of carcinoma |
Primary tumor grade | · Tumor grade (TNM and clinical stage) · Parameters of the primary tumor (size, number, invasiveness, histological variant, and characteristics) · Localization, size, and number of metastases (removed/affected), and capsule infiltration |
Genetics (tumor) | · BRAF, RET, NTRK, TERT, PAX8/PPAR-γ, RAS (mutation detection method, laboratory tests, and results) |
Instrumental diagnostics | Date · US · Endoscopy · X-ray Tumor dynamics assessment (RECIST): · CT/MRI (± contrasting) · SPECT(/CT) · PET/CT |
Surgery | Date of surgery MF · Surgical volume: on the thyroid gland, on regional lymph nodes (including ASL), outside the neck · Use of intraoperative neuromonitoring · Complications (surgical treatment): Date of registration · Postoperative laryngeal paresis/paralysis · Grade of postoperative hypoparathyroidism · Other (specify) |
Radioiodine diagnostics (RD) and radioiodine therapy (RT) | Date of procedure MF · Radioisotope: Iodine-123, Iodine-131, and Iodine-124 · Activity (MBq/mKi): automatic recalculation · SPECT(/CT) · PET/CT Distribution and accumulation of radiopharmaceuticals in projecting the thyroid bed, regional neck lymph nodes, retroperitoneal lymph nodes, lungs, bones, and other localizations of pathological accumulation The presence of radioiodine-resistant metastases (RR date to be registered), localization and size of foci, and progression (RECIST 1.1.) Complications (RT): · Postoperative laryngeal paresis/paralysis · Grade of postoperative hypoparathyroidism · Other |
Laboratory diagnostics | Date of analysis · TTH · FT (4) · TG · TgAb · CEA · Cyfra 21.1 · Proteinuria (AE) · Creatinine (AE) · AspAT (AE) · ALAT (AE) |
Laboratory diagnostics | · Bilirubin (AE) · Hemoglobin (AE) · Leukocytes (AE) · Neutrophils (AE) · Platelets (AE) |
Histology | Date of examination · # of examination (glass, blocks) · MF · Histological type and carcinoma variant · Size and localization of the dominant tumor in the thyroid · Presence and character of carcinoma intrinsic capsule · Invasion/infiltration of thyroid capsule carcinoma · Carcinoma multifocality in the thyroid · Angioinvasion in the thyroid · Number of removed ASL lymph nodes · Number of metastatic ASL lymph nodes |
Hormone therapy | Date of administration · Daily dose, µg |
Radioiodine resistance (summary) | RR date (see RD/RT section) Total activity of iodine-131 (including that at which RR was registered, GBq/mKi) |
Targeted therapy | Date (administration, correction, and withdrawal) · Drug (INN) · Daily dose, mg · Reasons for drug withdrawal |
Adverse events (complications) of targeted therapy | AE registration date · ECOG, st. · Arterial hypertension, st. · Diarrhea, st. · Nausea and vomiting, st. · Fatigue, st. · Decrease in body weight and loss of appetite, st. · Renal toxicity (proteinuria), st. · Rash (hand–foot syndrome), st. · Bleeding threat, st. · Liver toxicity (AspAT, ALAT, bilirubin), st. · Hypothyroidism, st. · Myelosuppression, st. |
Note: TTH, thyrotropic hormone; TG, thyroglobulin; TgAb, thyroglobulin antibodies; FT, free tetraiodothyroxine; ASL, anatomical and surgical level of regional lymph nodes; MF, medical facility; AE, adverse events.
This databank is an integration project for developing digital medicine to address the following healthcare challenges:
- A multidisciplinary paradigm of multimodal data collection and analysis unified for prompt and evidence-based patient care;
- Digital resource creation that is available to the interested doctor to record and analyze his/her own experience and prompt peer expert support in real clinical practice;
- An innovative digital platform for multicenter scientific and practical research;
- A matrix for clinical and epidemiological research, radiomics development, deep machine learning, and artificial intelligence in medicine;
- New RR-DTC case predictions and their verification and optimal treatment tactic selection;
- Evidence-based experience consolidation in the effective and safe treatment of patients (e.g., surgery, radioiodine therapy, hormone therapy, and targeted therapy).
ACTUAL CONTENT
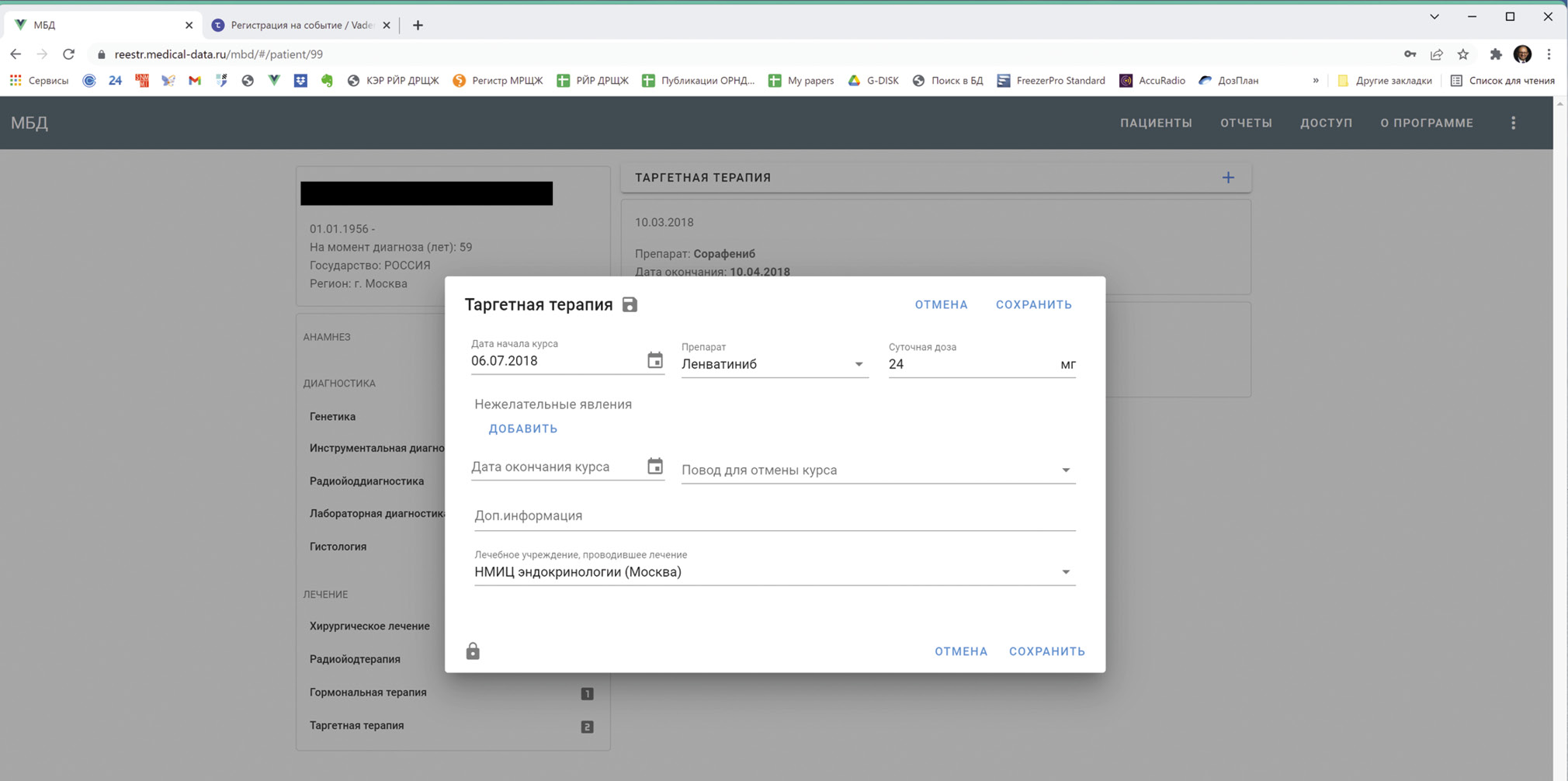
At present, the IDB RR-DTC project is already available at http://reestr.medical-data.ru/mbd/ for the general medical community both for review and participation. The accessible version is the result of the efforts of 11 doctors, covering the observation data of 228 patients from 48 regions of the Russian Federation. All doctors who treat patients with RR-DTC, including oncologists, surgeons, radiologists, nuclear medicine specialists, endocrinologists, chemotherapists, pathologists, and geneticists, are welcome to join the project. The developers offer to directly register in the system when entering the website. The IDB RR-DTC interface (Fig. 1) is easy-to-use for any doctor since the project was designed and implemented by a doctor.
Fig. 1. The interface of the Interdisciplinary Patient Databank Project for Radioiodine-Refractory Differentiated Thyroid Cancer (IDB RR-DTC).
Sections to be filled out correspond to the classic medical history and outpatient records. Doctors can broaden their professional horizons without leaving the workplace, learn teamwork, exchange experience in managing patients with RR-DTC, become involved in scientific and analytical work, and become coauthors of publications in both domestic and foreign scientific medical journals by participating in the project.
The multimodal data bank (clinical, laboratory, and radiological) creation develops and facilitates interdisciplinary collaboration, which, in turn, forms the basis for multicenter and national clinical and epidemiological evidence-based studies and the improvement of diagnostic, treatment, and rehabilitation technologies for patients with cancer. This is true for neuroendocrine tumors that are highly aggressive and refractory to standard disease treatment.
ADDITIONAL INFORMATION
Funding source. The publication is prepared with financial support of EISAI company. Authors are completely responsible for the content as well as reduction remarks.
Competing interest. Authors declare the absence of competing interests.
Authors’ contribution. Rumiantsev P.O. — idea, design, content of the article; Baidak A. — figures, technical assistance. All authors made a substantial contribution to the conception of the work, acquisition, analysis, interpretation of data for the work, drafting and revising the work, final approval of the version to be published and agree to be accountable for all aspects of the work.
Acknowledgements. The authors express their gratitude to Association of theranostics development, Group of clinics “SOGAZ-Medicine”, Association of endocrine surgeons, Russian society of specialists in head and neck tumors.
About the authors
Pavel O. Rumyantsev
International Medical Center “SOGAZ-MEDICINE”
Email: pavelrum@gmail.com
ORCID iD: 0000-0002-7721-634X
SPIN-code: 7085-7976
MD, Dr.Sci. (Med), Deputy Director of Oncoendocrine Institute
Russian Federation, 8, Malaya Konyushennaya, Saints Peterburg, 191186Andrey G. Baidak
M-Data
Author for correspondence.
Email: baidak@medical-data.ru
Russian Federation, Orel
References
- Chissov VI, Starinsky VV, Petrova GV. Malignant neoplasms in Russia since 2008 (morbidity and mortality). Moscow: P.A. Herzen Federal State Medical Research Institute of Rosmedtechnology; 2010. 256 p. (In Russ).
- Kaprin AD, Starinsky VV, Petrova GV. The state of oncological care for the population of Russia in 2019. Moscow: P.A. Herzen Moscow State Medical Research Institute — branch of the FSBI NMIC of Radiology of the Ministry of Health of Russia,2020. 236 p. (In Russ).
- Pacini F, Fuhrer D, Elisei R, et al. 2022 ETA Consensus Statement: what are the indications for post-surgical radioiodine therapy in differentiated thyroid cancer? Eur Thyroid J. 2022;11(1):e210046. doi: 10.1530/ETJ-21-0046
- Rumyantsev PO, Fomin DK, Rumyantseva UV. Criteria of resistance of highly differentiated thyroid cancer to therapy with radioactive iodine. Tumors Head Neck. 2014;(3):4–9. (In Russ).
- Rumyantsev PO, Slashchuk KY, Korenev SV, et al. Interdisciplinary data bank in oncoendocrinology. Medullary thyroid cancer and syndromes of multiple endocrine neoplasia type 2. Endocrine Sur. 2019;13(3):105–117. (In Russ). doi: 10.14341/serg11270
- Almazov AA, Rumyantsev PO, Kupreev PP, et al. Medical decision support systems; analysis of multimodal data, the difference between “human” and “machine” approaches, social problems of collection and turnover of biomedical data. Doctor Information Technol. 2020;(2):28–35. (In Russ). doi: 10.37690/1811-0193-2020-2-28-35
- Gatta G, van der Zwan JM, Casali PG, et al. Rare cancers are not so rare: the rare cancer burden in Europe. Eur J Cancer. 2011;47(17):2493–2511. doi: 10.1016/j.ejca.2011.08.008
- Filetti S, Durante C, Hartl D, et al. Thyroid cancer: ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann Oncol. 2019;30(12):1856–1883. doi: 10.1093/annonc/mdz400
- Clinical recommendations “Differentiated thyroid cancer”. Moscow; 2020. 47 p. (In Russ).
Supplementary files