“Superior Pectus Carinatum” (Currarino–Silverman Syndrome) in a 66-year-old woman: a case report
- Authors: Mannatrizio D.1, Fascia G.1, Guglielmi G.1,2
-
Affiliations:
- Department of Clinical and Experimental Medicine, Foggia University School of Medicine
- Radiology Unit, Barletta University Hospital
- Issue: Vol 3, No 2 (2022)
- Pages: 141-148
- Section: Case reports
- Submitted: 14.03.2022
- Accepted: 06.06.2022
- Published: 14.07.2022
- URL: https://jdigitaldiagnostics.com/DD/article/view/104865
- DOI: https://doi.org/10.17816/DD104865
- ID: 104865
Cite item
Abstract
The premature fusion of some of the sternal ossification centers and the obliteration of the manubrio-sternal joint caused a rare deformity called Currarino–Silverman syndrome. Patients present an abnormally short sternum with a forward angulation at the manubrio-sternal junction. Cardiopulmonary diseases and spinal deformities are the most frequent related disorders. It was also described as a component of Turner’s and Noonan’s syndromes.
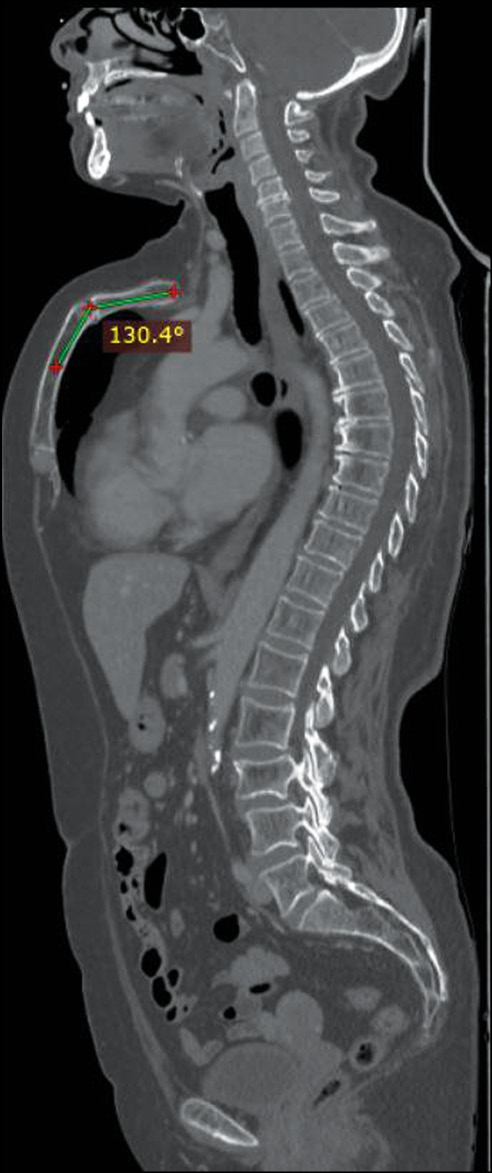
Herein, we present the case of a 66-year-old woman who presented to our clinic for follow-up computed tomography after surgery and chemotherapy for breast cancer with frequent episodes of dyspnea, wheezing, bronchitis, and mild dyspnea annually, which was more frequent during childhood. Computed tomography showed the absence of metastatic lesions and other accompanying diseases, except for a rare deformity of the anterior chest wall, the so-called, a “superior” pectus carinatum, a chondromanubrial deformity with a dorsal-open angle of 130º, and a sternum body length of 9 cm, which is not depressed in the lower third.
Keywords
Full Text
BACKGROUND
The most common congenital chest wall malformations (CWMs) are pectus excavatum (PE) and pectus carinatum (PC). These anomalies present during the first years of life and occur during childhood. PC is less frequent than PE, and it affects 5%–15% of all patients with CWMs and occurs in approximately 1:1000 to 1:10000 of all live births, with a male predominance (4:1) (1) (2). The mild forms are more frequent than the severe forms of PC (3). The chondromanubrial type was first described by Guido Currarino and Frederic Silverman in 1958 (4).
The real etiopathogenesis of type 2 PC is still unknown, but the most plausible hypothesis is that the deformity is caused by an excessive growth of costal cartilage secondary to genetic factors, which results in a sternal deformity without sternal pathology (5).
A short, solid sternum with prominent outward protrusion and bilateral deformity of the second to fifth costal cartilages that form an acute intercostal angle is the pathognomonic aspect of this syndrome (5). The xiphoid process is usually directed forward, even if it can be also absent. In very rare cases, a “superior or chondromanubrial PC” has a normal length, and the sternum is not depressed in the lower third.
Degenerative changes in hyaline cartilages, atypical fibrils, reduced number of chondrocytes, and thin periosteum are observed microscopically (6).
DESCRIPTION OF THE CASE
A 66-year-old woman presented to our clinic to undergo a follow-up computed tomography (CT) after surgery and chemotherapy for breast cancer. She also reported 3–4 episodes of bronchitis, wheezing, and mild dyspnea annually, which was more frequent during her childhood.
She underwent CT of the chest and abdomen with contrast enhancement, which showed the absence of metastatic lesions and any other respiratory tract disorders but revealed a chondromanubrial deformity with a dorsal-open angle of 130° and a sternum body length of 9 cm, which was not depressed in the lower third. Dorsal kyphosis was also present.
During the anamnesis, her CWM was not evident because she was dressed up with a turtleneck sweater that covered her chest.
She reported having an unknown inborn CWM, which was first noted at the age of 3 and progressed until the age of 13 years, accompanied by dyspnea and wheezing that accentuated during exercise and recurrent respiratory infections.
Moreover, the aesthetic appearance of her chest had psychological implications in her childhood, such as having strange feeling among her schoolmates, insecurity, withdrawal, and avoidance of all sports activities that require exposure of the chest. She has passed her childhood hiding her health problem.
DISCUSSION
The Currarino–Silverman (CS) syndrome is an extremely rare congenital deformity of the sternum.
It is also known by the terms “pouter pigeon chest,” “chondromanubrial deformity,” “type 2 pectus carinatum,” and “pectus arcuatum.” According to the original paper of Currarino and Silverman (4), this rare developmental anomaly is characterized by prominent sternal angulation with a decrease in length as a result of congenital complete non-segmentation or premature synostosis of the sternum.
The theory of a congenital etiology by gene downregulation is predominant (7-11). Essentially, a family history of CWMs is present in approximately 25% of the patients (1).
An abnormality of the differentiation of anterior segment mesenchymal cells and abnormal migration of mesenchymal cardiac precursors to the endothelial heart tube at the time of cardiogenesis may result in defects of endocardial cushion, sternum, and aortic arch derivatives (7). This syndrome is often combined with congenital heart defects and spinal abnormalities (kyphosis, scoliosis, and kyphoscoliosis), and it was also described as a component of Turner’s and Noonan’s syndromes (8).
Frequent confusion of CS syndrome with PE deformity is still an issue since one-third of patients present with concomitant mild to moderate depression of the lower third of the sternum (9).
Indeed, CS syndrome can be easily mistaken with PE, as both deformities appear in an almost similar fashion, but surgical approaches are very different. A distinctive feature of a PE deformity is the beginning of the sternal depression at the angle of Louis that becomes progressively deeper toward the xiphoid process with distorted and elongated cartilages (10). The angle of Louis must be less than 110° to be considered a true depression (5). Thus, confusion about classification is still an actual issue.
A uniform classification is a basis for surgical treatment and assessment of its short- and long-term results.
In 2006, Acastello classified CWMs based on the site of the defect’s origin (type 1, cartilaginous; type 2, costal; type 3, chondrocostal; type 4, sternal; type 5, clavicle-scapular) and attributed CS syndrome to the cartilaginous type 2 (superior) PC. Further, Torre et al. distinguished superior PC into two types:
- Type 1: The “inferior” or “chondrogladiolar” is the most frequent type, in which the sternal protrusion is located in the inferior or mid sternum and the last ribs can be slightly or severely depressed on lateral aspects.
- Type 2: The “superior” or “chondromanubrial” is the less frequent type, which has been further divided in two types depending on the external aspect. The first type is characterized by a superior PC with an inferior PE in which the sternum is S-shaped on a lateral view. This anomaly is classified as cartilaginous anomalies and called type II. Although this anomaly is considered a cartilaginous anomaly and a type II PC in the Acastello classification, Torre et al. classified it as part of sternal anomalies because of the sternal origin of the anomaly. The second type is a “superior PC” without the typical features of the CS syndrome. The sternum has a normal length and is not depressed in the lower third. This anomaly is probably due to a cartilage anomaly similarly to inferior PC. Torre et al. proposed to use the term “superior PC” only for this type and to include this anomaly in the first category (cartilaginous anomalies) of CWM classification. This entity is extremely rare.
Fig 1. Sagittal whole body computed tomography scan showing an arching sternum and a chondromanubrial deformity with a dorsal-open angle of 130°.
Fig 2. Sagittal whole body computed tomography scan showing a sternum body length of 9 cm and dorsal kyphosis.
Most patients with CS syndrome are asymptomatic; therefore, surgical correction is optional, and different opinions exist about the ideal age for surgical correction. Asthma and chronic bronchitis, which occur in 16% of the patients, are the most common associated concomitant diseases, responsible for bronchial and pulmonary symptoms (11).
Fig 3. Lateral plain radiograph appearance of a type 2 pectus carinatum.
Fig 4. Computed tomography reconstruction of the rib cage showing that the sternum has a normal length and is not depressed in the lower third.
Kyphosis is present, to a greater or lesser degree, in almost all patients. Pain or tenderness at the protrusion site, decreased endurance, or palpitations can be present. Limitations at work and in sports and underachievement in school, in the absence of respiratory and heart diseases, should be attributed to emotional alterations.
A strong correlation of inborn cardiac pathology with CS syndrome has been reported, such as VSD, patent ductus arteriosus, atrial septal defect, tetralogy of Fallot, transposition of the great arteries, and coarctation of the aorta.
Chest CT with three-dimensional reconstruction, or magnetic resonance imaging in children with altered tissue density and radiation concern, is the best preoperative imaging for the evaluation of patients with CS. It allows the differential diagnosis with other pectus deformities, determination of the exact angles of the costal cartilages to the sternum, and enhances surgical planning.
In comparison to PE, several less invasive techniques have been developed for the correction of PC, including the Abramson procedure and its modifications, along with nonsurgical options such as observation, orthotic bracing, and dynamic compression (2) (12). However, due to the extreme rarity of the disease, challenging deformity, and variable anatomy of a fused sternum, there are no clear guidelines in the treatment approaches.
This unique growth pattern contributes to an almost universal failure of conservative treatment options, such as the vacuum bell or a compressive orthosis. The best surgical option remains the relatively aggressive Ravitch-type procedure with multi-level wedge osteotomy (12) (13), allowing for the achievement of a satisfactory outcome.
Given the rarity of the deformity, surgical correction should be completed by a multidisciplinary team, including thoracic reconstructive surgeons with experience in pectus deformities. The preferred age for correction is late puberty or adulthood, as cartilage resection will be performed when the rib growth ends (1) (14) (15). It is important to keep in mind the potential for thoracic dystrophy should cartilage resection be performed at a young age or too extensively. Thus, other studies have reported the preferable age of 5–7 years or early adolescence (5) (16). Some patients who refused to undergo surgery turned to bodybuilding to improve and define the musculature around the CWM to minimize the appearance of protrusion. Even if this approach does not correct the abnormality, it can improve self-esteem and confidence. For female patients, one possibility to make protrusion less noticeable and improve the appearance of the chest is breast augmentation (17).
CONCLUSION
Although a rare condition, CS can be easily diagnosed through its typical radiological findings. Therefore, clinicians must be aware of and recognize this imaging pattern to make an accurate diagnosis and prevent further examination and aggressive treatment.
ADDITIONAL INFORMATION
Funding source. This study was not supported by any external sources of funding.
Competing interests. The authors declare that they have no competing interests.
Authors’ contribution. All authors made a substantial contribution to the conception of the work, acquisition, analysis, interpretation of data for the work, drafting and revising the work, final approval of the version to be published and agree to be accountable for all aspects of the work.
Consent for publication. Written consent was obtained from the patient for publication of relevant medical information within the manuscript.
About the authors
Domenico Mannatrizio
Department of Clinical and Experimental Medicine, Foggia University School of Medicine
Email: dr.mannatrizio@gmail.com
ORCID iD: 0000-0003-3365-7132
MD
Italy, FoggiaGiacomo Fascia
Department of Clinical and Experimental Medicine, Foggia University School of Medicine
Email: giacomo.fascia@unifg.it
ORCID iD: 0000-0001-5244-5093
MD
Italy, FoggiaGiuseppe Guglielmi
Department of Clinical and Experimental Medicine, Foggia University School of Medicine; Radiology Unit, Barletta University Hospital
Author for correspondence.
Email: giuseppe.guglielmi@unifg.it
ORCID iD: 0000-0002-4325-8330
MD, Professor
Italy, Foggia; BarlettaReferences
- Shamberger RC, Welch KJ. Surgical correction of pectus carinatum. J Pediatr Surg. 1987;22(1):48–53. doi: 10.1016/s0022-3468(87)80014-3
- Muntean A, Stoica I, Saxena AK. Pigeon chest: comparative analysis of surgical techniques in minimal access repair of pectus carinatum (MARPC). World J Pediatr. 2018;14(1):18–25. doi: 10.1007/s12519-018-0121-2
- Emil S. Current options for the treatment of pectus carinatum: when to brace and when to operate? Eur J. 2018;28(4):347–354. doi: 10.1055/s-0038-1667297
- Currarino G, Silverman F. Premature obliteration of the sternal sutures and pigeon-breast deformity. Radiology. 1958;70(4):532–540. doi: 10.1148/70.4.532
- Fokin A, Steuerwald NM, Ahrens WA, Allen KE. Anatomical, histologic, and genetic characteristics of congenital chest wall deformities. Semin Thorac Cardiovasc Surg. 2009;21(1):44–57. doi: 10.1053/j.semtcvs.2009.03.001
- Fokin A. Pouter pigeon breast. Chest Surg Clin N Am. 2000;10(2):377–391.
- Gabrielsen T, Ladyman G. Early closure of the sternal sutures and congenital heart disease. Am J Roentgenol Radium Ther Nucl Med. 1963;89:975–983.
- Chidambaram B, Mehta AV. Currarino-Silverman syndrome (pectus carinatum type 2 deformity) and mitral valve disease. Chest. 1992;102(3):780–782. doi: 10.1378/chest.102.3.780
- Regier DS, Oetgen M, Tanpaiboon P. Mucopolysaccharidosis type IVA. 2013 Jul 11 [updated 2021 Jun 17]. In: Adam MP, Ardinger HH, Pagon RA, et al., ed. GeneReviews [Internet]. Seattle (WA): University of Washington, Seattle; 1993–2022.
- Martinez-Ferro M, Bellia-Munzon G, Schewitz IA, Toselli L. Pectus carinatum: when less is more. Afr J Thorac Crit Care Med. 2019;25(3):10.7196/AJTCCM.2019.v25i3.019. doi: 10.7196/AJTCCM.2019.v25i3.019
- Lester C. Pigeon breast (pectus carinatum) and other protrusion deformities of the chest of developmental origin. Ann Surg. 137(4):482–489. doi: 10.1097/00000658-195304000-00008
- Welch KJ, Vos A. Surgical correction of pectus carinatum (pigeon breast). J Pediatr Surg. 1973; 8(5):659–667. doi: 10.1016/0022-3468(73)90404-1
- Coelho MS, Santos A, Pizarro L, et al. “Pectus excavatum/pectus carinatum”: tratamento cirúrgico. J Pneumol. 1983;10(Supрl):47.
- Ramadan S, Wilde J, Tabard-Fougère A, et al. Cardiopulmonary function in adolescent patients with pectus excavatum or carinatum. BMJ Open Respir Res. 2021;8(1):e001020. doi: 10.1136/bmjresp-2021-001020
- Buziashvili D, Gopman JM, Weissler H, et al. An evidence-based approach to management of pectus excavatum and carinatum. Ann Plast Surg. 2019;82(3):352–358. doi: 10.1097/SAP.0000000000001654
- Szafer D, Taylor JS, Pei A, et al. A simplified method for three-dimensional optical imaging and measurement of patients with chest wall deformities. J Laparoendosc Adv Surg Tech A. 2019;29(2):267–271. doi: 10.1089/lap.2018.0191
- Geraedts TC, Daemen JH, Vissers YL, et al. Minimally invasive repair of pectus carinatum by the Abramson method: a systematic review. J Pediatr Surg. 2021;5:S0022-3468(21)00829-0. doi: 10.1016/j.jpedsurg.2021.11.028
- Brichon PY, Wihlm JM. Correction of a severe pouter pigeon breast by triple sternal osteotomy with a novel titanium rib bridge fixation. Ann Thorac Surg. 2010;90(6):e97–99. doi: 10.1016/j.athoracsur.2010.08.068
- Tarhan T, Meurer A, Tarhan O. Combined extra-/intrathoracic correction of pectus carinatum and other asymmetric chest wall deformities: A novel technique. Oper Orthop Traumatol. 2018;30(6):469–478. doi: 10.1007/s00064-018-0567-3
- McHam B, Winkler L. Pectus Carinatum. 2021 Aug 9. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2022.
- Rea G, Sezen CB. Chest wall deformities. 2021 Aug 11. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2022.
- Ramadan S, Wilde J, Tabard-Fougère A, et al. Cardiopulmonary function in adolescent patients with pectus excavatum or carinatum. BMJ. 2021;8(1):e001020. doi: 10.1136/bmjresp-2021-001020
Supplementary files