Rare localization of avascular necrosis during treatment of COVID-19 with glucocorticosteroids
- Authors: Gonchar A.P.1, Blokhin I.A.1, Shumskaya Y.F.1,2
-
Affiliations:
- Research and Practical Clinical Center for Diagnostics and Telemedicine Technologies
- The First Sechenov Moscow State Medical University (Sechenov University)
- Issue: Vol 3, No 4 (2022)
- Pages: 384-392
- Section: Case reports
- Submitted: 31.08.2022
- Accepted: 11.11.2022
- Published: 30.12.2022
- URL: https://jdigitaldiagnostics.com/DD/article/view/110718
- DOI: https://doi.org/10.17816/DD110718
- ID: 110718
Cite item
Abstract
The development of bony avascular necrosis induced by glucocorticoid treatment of COVID-19 is a common adverse effect, with femoral head being the most commonly affected. Timely detection of avascular necrosis is important in the prevention of osteoarthrosis and other complications.
We present a clinical case of a 54-year-old patient hospitalized for novel coronavirus infection with complaints of severe pain in both knees 2 weeks after the disease onset. Magnetic resonance imaging revealed pronounced changes in both knees, corresponding to avascular necrosis. The results of conservative therapy, including non-steroidal anti-inflammatory drugs and bisphosphonate bone resorption inhibitors, produced a pronounced positive result. At follow-up examination 3 months later, there was no pain, but the knee joints still had slight restrictions of movement. Magnetic resonance imaging showed a significant decrease in the previously detected changes.
The side effects of glucocorticoids (impaired glucose tolerance, increased blood pressure, tachycardia, gastrointestinal erosive ulcers, sleep disorders, etc.) are widely known, but knee osteonecrosis caused by steroid intake rarely comes to the attention of clinicians. This clinical case emphasizes the complex nature of osteonecrosis pathogenesis and demonstrates a wide range of complications in corticosteroid therapy.
Full Text
BACKGROUND
The novel coronavirus infection, discovered in Wuhan, China, in December 2019, has triggered a global pandemic. The use of glucocorticosteroids (GCSs) to treat novel coronavirus infection is pathogenetically justified and widespread [1]. However, even a single dose of GCS can cause the development of avascular necrosis [2]. The literature describes several clinical cases of femoral head avascular necrosis in patients treated with GCS [3, 4]. However, the cases of osteonecrosis in other areas, particularly the knee joints, are mentioned much less frequently [5]. Early diagnosis of this pathology is critical for preventing arthritis and other complications [6].
This paper describes a clinical case of avascular necrosis of the bone structures of both knee joints that developed while treating coronavirus infection with GCS.
CLINICAL CASE
Patient
A 54-year-old woman with complaints of severe cough and fever up to 39.5 °C for 6 days was hospitalized with the novel coronavirus infection. A chest CT revealed lung damage of >30%. The PCR test for SARS-CoV-2 RNA was positive. The medical history was insignificant.
During the hospital stay, the patient received parenteral dexamethasone at a daily dose of 20 mg for 5 days, followed by a 2-day break and subsequent reintroduction of dexamethasone at a daily dose of 12 mg for 5 days. Additionally, the patient received anticoagulant and antisecretory therapy.
On Day 15 of the disease, the patient experienced severe pain and substantial limitation of knee joint movement, which persisted at night. The knee joints were nontender on palpation. On Day 17 of the disease, the patient was discharged because of positive changes. Nonsteroidal anti-inflammatory drugs (NSAIDs) were recommended for treating knee joint pain.
Magnetic resonance imaging (MRI) of both knee joints was performed 1.5 months after hospitalization due to persistent pain and limited movement in the knee joints.
Instrumental findings
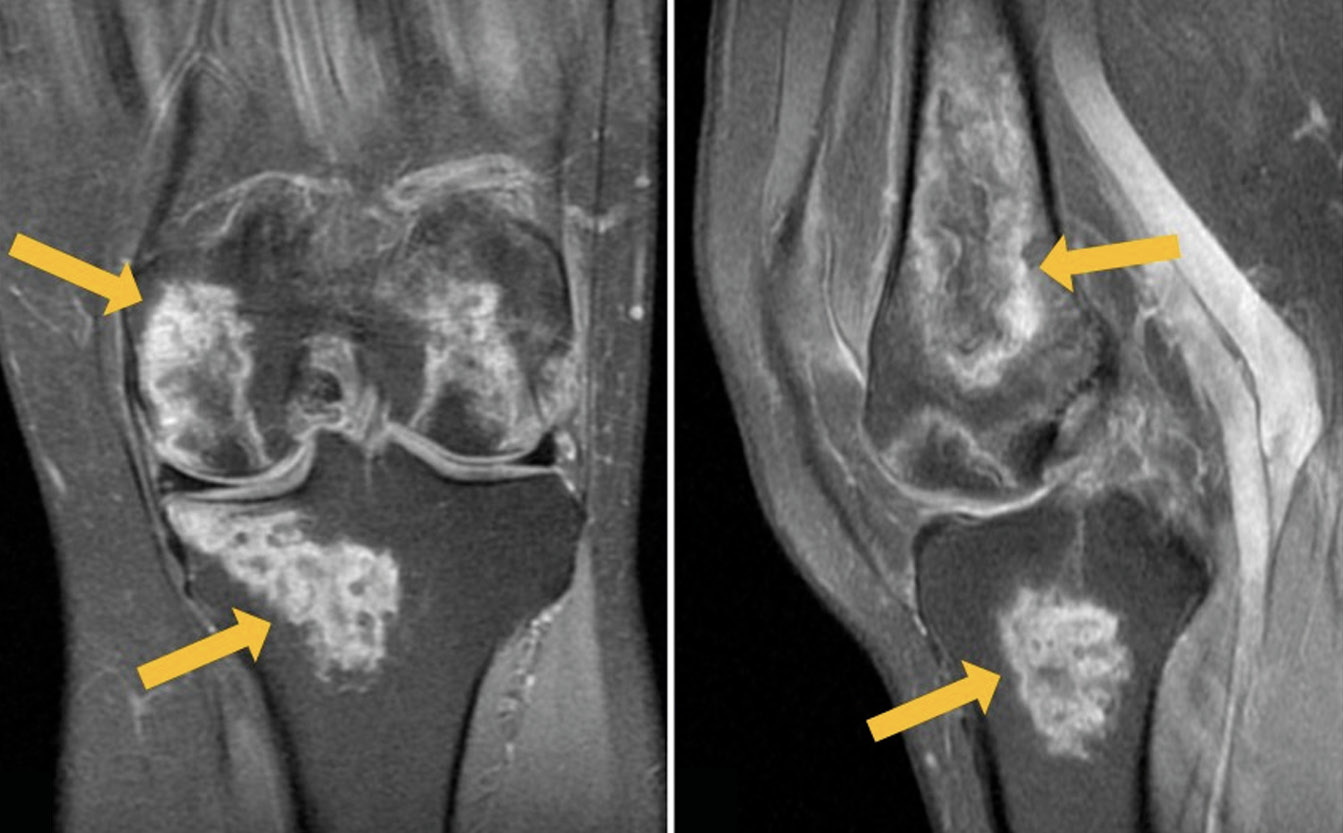
MRI of the left knee joint: lesions in the distal parts of the femoral diaphysis and femoral condyles (with involvement of the articular surface) as well as in the patella, inhomogeneously hyperintense on PD-weighted (proton-weighted) images with fat suppression and hypo-/isointense on T1-weighted images (T1WI), with an irregular (“geographic”) shape and yellow marrow signal areas visualized in the central parts (Fig. 1). MRI of the right knee joint: similar lesions of the bone marrow of both femoral condyles, with involvement of the distal metaepiphysis and articular surface of the lateral condyle as well as the patella. A perilesional “double line” sign is visualized over a short distance for some lesions (Fig. 2).
Fig. 1. Primary MRI of the left knee joint: PDWI with fat suppression in the coronal (а) and sagittal (b) plane and T1WI in the sagittal plane (с). The arrows indicate areas of bone marrow edema in the form of a heterogeneous, irregularly shaped (“geographic”) MRI signal of femoral and tibial condyles.
Fig. 2. Primary MRI of the right knee joint: PDWI with fat suppression in the coronal (а) and sagittal (b) plane and T1WI in the sagittal plane (с). Thick arrows indicate areas of bone marrow edema in the form of a heterogeneous, irregularly shaped (“geographic”) MRI signal of femoral condyles and patella; thin arrows indicate the “double line” sign in the form of internal hyperintense (granulation tissue) and external hypointense (osteosclerosis) lines on PDWI.
The following diagnosis was made on the basis of medical history and identified MRI changes: “Avascular necrosis of the bone structures of both knee joints.”
Diagnosis and treatment
On the basis of MRI findings, medical history, and clinical pattern, the patient was diagnosed with avascular necrosis of the femoral and tibial condyles of both knee joints. Thus, the following conservative therapy (physiotherapy) was prescribed: magnet therapy and phonophoresis with NSAID-containing gel, NSAID therapy (tablets) for pain (as needed), vitamin D preparations, and bisphosphonate bone resorption inhibitors.
The therapy resulted in substantial positive changes after 3 months: The pain had subsided, but some limitations in knee joint movement remained (the patient could not do a deep squat).
A follow-up MRI of both knee joints revealed positive changes: Previously identified lesions had become substantially less severe (Figs. 3 and 4).
Fig. 3. Follow-up MRI of the left knee joint: PDWI with fat suppression in the coronal (а) and sagittal (b) plane and T1WI in the sagittal plane (с). Thick arrows indicate areas of bone marrow edema in the form of a heterogeneous, irregularly shaped (“geographic”) MRI signal of femoral condyles and patella; the thin arrow indicates the “double line” sign in the form of internal hyperintense (granulation tissue) and external hypointense (osteosclerosis) lines on PDWI.
Fig. 4. Follow-up MRI of the right knee joint: PDWI with fat suppression in the coronal (а) and sagittal (b) plane and T1WI in the sagittal plane (с). Thick arrows indicate areas of bone marrow edema in the form of a heterogeneous, irregularly shaped (“geographic”) MRI signal of femoral condyles and patella; thin arrows indicate the “double line” sign in the form of internal hyperintense (granulation tissue) and external hypointense (osteosclerosis) lines on PDWI.
DISCUSSION
The prevalence of osteonecrosis in patients with the novel coronavirus infection ranges from 5% to 58% [7]. Damage is more common to the femoral head than to the bone structures of the knee joints and other bones.
Secondary osteonecrosis of knee joint bone structures, particularly avascular necrosis, most commonly affects both femoral condyles, as in the clinical case presented here. Conversely, primary osteonecrosis affects only one of the condyles [8].
Avascular necrosis can develop as a result of GCS treatment, kidney disease, or hematological diseases. Some authors believe that drugs used to treat coronavirus infection, such as lopinavir and ritonavir, can contribute to the development of osteonecrosis [9]. In our case, this pathology most likely developed as a result of GCS treatment for coronavirus infection.
GCSs, which play an important role in treating novel coronavirus infection, are an independent risk factor for developing avascular necrosis. At the same time, the pathogenesis of osteonecrosis in these patients is unclear: In addition to GCS therapy, independent causes may include vascular thrombosis, adipocyte hypertrophy, fat embolism, hypercoagulopathy, endothelial destruction, and leukocyte aggregation [7, 10]. However, we could not find clinical cases of osteonecrosis due to the above factors in the literature [7, 9].
No agreement has been made on the duration of corticosteroid therapy or the dosage that increases the risk of osteonecrosis. Nonetheless, numerous studies suggest that controlling the cumulative dose of GCS is important in developing this pathology [11]. Thus, osteonecrosis of the bone structures of the knee joint was found to develop with a cumulative dose of prednisolone ranging from 1.012 to 6.562 g, [12, 13] whereas in other clinical cases, the cumulative dose of prednisolone was in the range of 0.9–1.413 g, with an average value of 1.156 g [14]. In our case, dexamethasone was used at a dose of 20 mg/day, followed by a decrease to 12 mg/day.
Agarwala et al. [14] describe a case of avascular necrosis in a 20-year-old woman after using methylprednisolone for 15 days. The patient experienced pain in the knees on Day 25 of the disease, with lesions of both condyles and the patella according to MRI. The same authors describe a case of a 16-year-old boy who developed pain in both hip joints and the right knee joint 4 months after the novel coronavirus infection when treated with methylprednisolone and dexamethasone for 19 days. In our clinical case, dexamethasone was used for 10 days with a 2-day break. The first complaints of knee pain occurred on Day 15 of the disease (Day 9 of GCS therapy), similar to the case of the abovementioned female patient, although our patient’s age differed considerably.
Another clinical case refers to avascular necrosis of the right knee joint in a 78-year-old woman with a history of bilateral gonarthrosis, more pronounced on the left, as well as concomitant cardiovascular diseases and obesity [15]. The treatment included antibacterial drugs, hydroxychloroquine, antiviral drugs (lopinavir, ritonavir), and oxygen therapy. Two weeks after discharge, the patient reported that the pain in the right knee joint had worsened. During this period, the patient also received GCS therapy for 9 days for bronchospasm. Seven days later, the patient developed local edema of the right knee joint. MRI revealed osteonecrosis of the right femoral medial condyle. GCS therapy cannot be considered the sole cause of avascular necrosis in this case; the presence of concomitant diseases is also a risk factor for developing osteonecrosis. However, the brevity between coronavirus infection and developing avascular necrosis suggests an effect of GCS therapy. In our clinical case, the patient had no other risk factors for developing avascular necrosis other than taking GCS; however, as in the example above, arthralgia developed rather quickly during GCS therapy.
Conversely, the study by Sulewski et al. [16] indicates insufficient evidence of the direct effect of GCSs on the development of osteonecrosis. This study analyzed 10 patients with confirmed coronavirus infection and signs of avascular necrosis. The mean age of the patients was 61 years. Although only four out of ten patients received GCSs, all of them developed avascular necrosis on Day 14 of the disease, on average. Li et al. [17] obtained similar data in a meta-analysis, confirming the theory of multifactorial pathogenesis of avascular necrosis in patients with the novel coronavirus infection. A deficiency of angiotensin-converting enzyme 2, which can cause bone destruction as well as vascular thromboses, as in the case of osteonecrosis development on GCS therapy, is a possible factor in the development of avascular necrosis in such patients. Thus, no clear consensus exists regarding the etiology and mechanism of avascular necrosis development in patients with novel coronavirus infection.
In our clinical case and cases presented by foreign authors, the treatment of avascular necrosis is primarily conservative, with the main goals of relieving pain, slowing the progression of osteonecrosis, and preventing fractures and arthritis. At the same time, no generally accepted treatment scheme is available [18]. In turn, some researchers confirm that combination therapy with bisphosphonates is effective for treating osteonecrosis, including in the early stages, as our clinical case demonstrates [19, 20].
Thus, early identification of patients at high risk of developing avascular necrosis because of the novel coronavirus infection is critical in preventing arthritis and other complications.
CONCLUSION
We present a clinical case of MRI-detected bilateral avascular necrosis of the bone structures of the knee joint during treatment of COVID-19 with GCSs. GCS therapy has well-known side effects, such as impaired glucose tolerance, increased blood pressure, tachycardia, erosive and ulcerative lesions of the gastrointestinal tract, and sleep disturbances. However, avascular necrosis of the bone structures of the knee joints caused by GCS therapy is rarely brought to the attention of clinicians. Our case not only highlights the complexities of the pathogenesis of osteonecrosis but also demonstrates the wide range of complications associated with GCS therapy.
ADDITIONAL INFORMATION
Funding source. This article was not supported by any external sources of funding.
Competing interests. The authors declare that they have no competing interests.
Authors’ contribution. All authors made a substantial contribution to the conception of the work, acquisition, analysis, interpretation of data for the work, drafting and revising the work, final approval of the version to be published and agree to be accountable for all aspects of the work. A.P. Gonchar — concept and design of the paper; A.P. Gonchar — data collection and analysis; Yu.F. Shumskaya — data interpretation; I.A. Blokhin — approval of the final version of the paper.
Consent for publication. Written consent was obtained from the patient for publication of relevant medical information within the manuscript in Digital Diagnostics Journal.
Acknowledgments. The authors are thankful to Victoria Alexandrovna Matkova for her contribution to the publication.
About the authors
Anna P. Gonchar
Research and Practical Clinical Center for Diagnostics and Telemedicine Technologies
Email: a.gonchar@npcmr.ru
ORCID iD: 0000-0001-5161-6540
SPIN-code: 3513-9531
Russian Federation, Moscow
Ivan A. Blokhin
Research and Practical Clinical Center for Diagnostics and Telemedicine Technologies
Email: i.blokhin@npcmr.ru
ORCID iD: 0000-0002-2681-9378
SPIN-code: 3306-1387
Russian Federation, Moscow
Yuliya F. Shumskaya
Research and Practical Clinical Center for Diagnostics and Telemedicine Technologies; The First Sechenov Moscow State Medical University (Sechenov University)
Author for correspondence.
Email: yu.shumskaia@npcmr.ru
ORCID iD: 0000-0002-8521-4045
SPIN-code: 3164-5518
Russian Federation, Moscow; Moscow
References
- Temporary guidelines «Prevention, diagnosis and treatment of new coronavirus infection (COVID-19)». Version 3 (March 3, 2020) (approved by the Ministry of Health of the Russian Federation). (In Russ). Available from: https://www.garant.ru/products/ipo/prime/doc/73647088/. Accessed: 15.07.2022.
- Ali RS, Al-Sudani H, Tan IJ. Osteonecrosis of bilateral distal femurs in a pregnant patient following antenatal betamethasone. Cureus. 2022;14(3):e22735. doi: 10.7759/cureus.22735
- Zhang S, Wang C, Shi L, Xue Q. Beware of steroid-induced avascular necrosis of the femoral head in the treatment of COVID-19-experience and lessons from the SARS epidemic. Drug Des Devel Ther. 2021;15:983–995. doi: 10.2147/DDDT.S298691
- Agarwala SR, Vijayvargiya M, Pandey P. Avascular necrosis as a part of ‘long COVID-19’. BMJ Case Rep. 2021;14(7):e242101. doi: 10.1136/bcr-2021-242101
- Takao M, Sugano N, Nishii T, et al. Spontaneous regression of steroid-related osteonecrosis of the knee. Clin Orthop Relat Res. 2006;452:210–215. doi: 10.1097/01.blo.0000229278.51323.08
- Hines JT, Jo WL, Cui Q, et al. Osteonecrosis of the femoral head: an updated review of arco on pathogenesis, staging and treatment. J Korean Med Sci. 2021;36(24):e177. doi: 10.3346/jkms.2021.36.e177
- Disser NP, De Micheli AJ, Schonk MM, et al. Musculoskeletal consequences of COVID-19. J Bone Joint Surg Am. 2020;102(14):1197–1204. doi: 10.2106/JBJS.20.00847
- Karim AR, Cherian JJ, Jauregui JJ, et al. Osteonecrosis of the knee: review. Ann Transl Med. 2015;3(1):6. doi: 10.3978/j.issn.2305-5839.2014.11.13
- Patel MS, Gutman MJ, Abboud JA. Orthopaedic considerations following COVID-19: lessons from the 2003 SARS outbreak. JBJS Rev. 2020;8(7):e2000052. doi: 10.2106/JBJS.RVW.20.00052
- Kerachian MA, Séguin C, Harvey EJ. Glucocorticoids in osteonecrosis of the femoral head: a new understanding of the mechanisms of action. J Steroid Biochem Mol Biol. 2009;114(3-5):121–128. doi: 10.1016/j.jsbmb.2009.02.007
- Chan KL, Mok CC. Glucocorticoid-induced avascular bone necrosis: diagnosis and management. Open Orthop J. 2012;6:449–457. doi: 10.2174/1874325001206010449
- Powell C, Chang C, Naguwa SM, et al. Steroid induced osteonecrosis: an analysis of steroid dosing risk. Autoimmun Rev. 2010;9(11):721–743. doi: 10.1016/j.autrev.2010.06.007
- Takeda H, Nishise S, Fujishima S, et al. Osteonecrosis of the lateral femoral condyle in a patient with ulcerative colitis: report of a case. Clin J Gastroenterol. 2008;1(3):93–96. doi: 10.1007/s12328-008-0015-2
- Agarwala SR, Vijayvargiya M, Sawant T. Secondary osteonecrosis of the knee as a part of long COVID-19 syndrome: a case series. BMJ Case Rep. 2022;15(3):e248583. doi: 10.1136/bcr-2021-248583
- Angulo-Ardoy M, Ureña-Aguilera Á. Knee osteonecrosis after COVID-19. Fam Pract. 2021;38(Suppl 1):i45–i47. doi: 10.1093/fampra/cmab063
- Sulewski A, Sieroń D, Szyluk K, et al. Avascular necrosis bone complication after active COVID-19 infection: preliminary results. Medicina (Kaunas). 2021;57(12):1311. doi: 10.3390/medicina57121311
- Li W, Huang Z, Tan B, et al. General recommendation for assessment and management on the risk of glucocorticoid-induced osteonecrosis in patients with COVID-19. J Orthop Translat. 2021;31:1–9. doi: 10.1016/j.jot.2021.09.005
- Mont MA, Baumgarten KM, Rifai A, et al. Atraumatic osteonecrosis of the knee. J Bone Joint Surg Am. 2000;82(9):1279–1290. doi: 10.2106/00004623-200009000-00008
- Agarwala S, Sharoff L, Jagani N. Effect of zoledronic acid and alendronate on bone edema and pain in spontaneous osteonecrosis of the knee: a new paradigm in the medical management. Rev Bras Ortop (Sao Paulo). 2020;55(5):543–550. doi: 10.1016/j.rboe.2017.12.008
- Jureus J, Lindstrand A, Geijer M, et al. Treatment of spontaneous osteonecrosis of the knee (SPONK) by a bisphosphonate. Acta Orthop. 2012;83(5):511–514. doi: 10.3109/17453674.2012.729184
Supplementary files